Visible Results in 4 Weeks: Clinical Efficacy Testing for Premium Eye Care Brands
A rigorous B2B framework for validating under-eye claims, protecting compliance, and converting premium positioning into defensible market proof.
This white paper explains how premium eye care brands can substantiate fast-acting claims with clinical rigor, manufacturable precision, and documentation strong enough for global retail, marketplace, and regulatory scrutiny.
Market Intelligence: Why Eye Care Claims Fail or Scale
In premium eye care, the market does not reward vague promises. Buyers, distributors, and compliance teams want a claim that can survive a hostile audit, a skeptical retailer review, and a social-media comparison test.
The phrase “visible results in 4 weeks” is commercially powerful, but it is only asset-grade when the claim is supported by a protocol that controls lighting, image capture distance, baseline variability, usage compliance, and statistical significance. Without those controls, the claim becomes a liability rather than a differentiator.
For B2B buyers, the real question is not whether a serum or cream feels elegant. The real question is whether the formula, packaging, manufacturing process, and clinical dossier can jointly support a repeatable outcome under real distribution pressure.
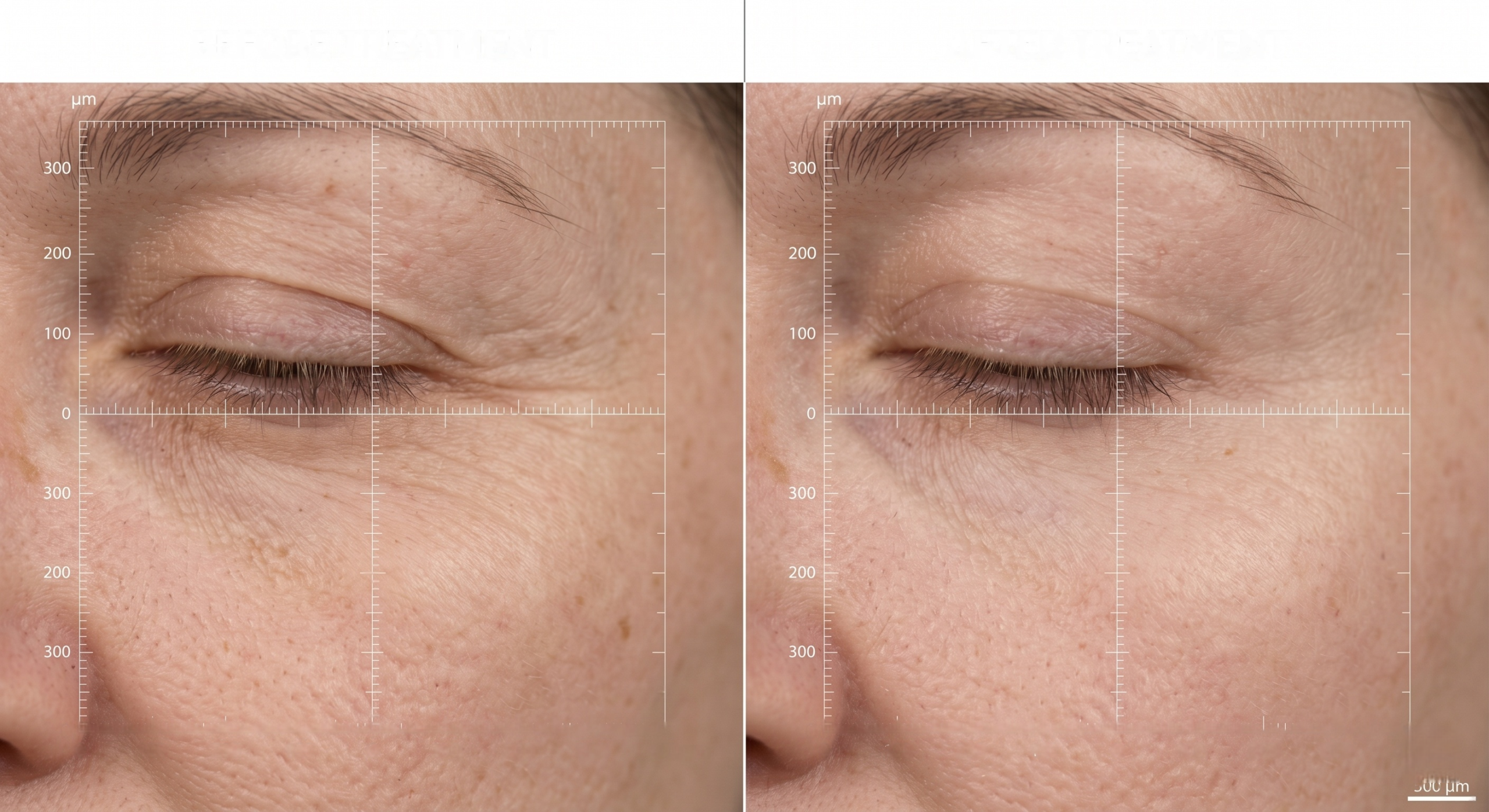
Clinical Efficacy Is Not Marketing Language; It Is a Controlled Evidence System
Clinical efficacy trials ask a narrower and more demanding question than consumer testimonials: does the product produce the expected result under predefined, controlled conditions? The distinction between efficacy and effectiveness is not semantic; it is the difference between a claim that can be defended and a claim that can be challenged.
As the NCBI Bookshelf review on efficacy versus effectiveness trials explains, efficacy evaluates the expected result under ideal conditions, while effectiveness measures real-world benefit. Premium eye care brands must bridge both if they want a claim that works in marketing and in procurement review.
That bridge starts with endpoint design. For under-eye products, the best endpoints are usually a combination of image-based wrinkle depth, puffiness reduction, color uniformity, hydration shift, and expert grading on a standardized scale. If the product also targets barrier support, the protocol may include corneometry or transepidermal water loss, provided the study population and instrumentation are appropriate.
In a credible 4-week eye-care protocol, the study should define the baseline condition, the usage frequency, the imaging device, the timepoints, and the statistical plan before first enrollment. The result should be a dossier that is repeatable, not a one-off win.
Why the 4-Week Window Matters
Four weeks is commercially useful because it is long enough to capture early biologic changes and short enough to align with product sampling cycles, creator campaigns, and retail launch calendars. For under-eye care, this window can be sufficient to show visible changes in hydration, surface smoothness, and appearance of fatigue, especially when the formulation uses humectants, peptides, caffeine, film-formers, or barrier-supportive lipids.
It is also short enough to reduce dropout and inventory risk, which matters for brands running low-MOQ validation through a micro-batch system. A 50-unit pilot can test a concept, collect proof, and determine whether to scale before capital is locked into a large production commitment.
The Mechanistic Logic Behind Visible Eye-Care Results
Premium eye care products typically work through one or more of five biological levers: hydration replenishment, barrier reinforcement, microcirculation support, decongestive signaling, and surface optical smoothing. Each lever can produce a visible benefit, but each also requires a different validation strategy.
Hydration-focused products often use glycerin, hyaluronic acid, betaine, and polyols to alter the stratum corneum water balance. If the formula also contains occlusives or emulsifiers that reduce transepidermal water loss, the visible improvement may emerge as reduced fine-line prominence and a more supple under-eye texture.
De-puffing systems often rely on caffeine or botanical vasomodulators, but claims must be careful. If the result is transient, the study should be designed to detect short-term appearance changes, not long-term structural transformation that the formula cannot actually deliver.
Optical smoothing systems use film-forming polymers or soft-focus ingredients to improve immediate surface reflectance. In those cases, the claim must be framed as visible cosmetic improvement rather than permanent anatomical change.

Technical Deep-Dive: How to Prove “Visible Results in 4 Weeks” Without Overclaiming
When brands say “visible results,” they are making a consumer-facing promise that must still map to measurable endpoints. The best programs convert an aesthetic promise into a technical framework with controlled photography, expert grading, and predefined statistical thresholds.
The first step is to define the benefit class. In eye care, that class may include reduction in visible fine lines, decrease in under-eye puffiness, improvement in dark-circle appearance, better skin feel, and hydration enhancement. Each benefit class should be supported by a different or complementary measurement system, because one endpoint rarely captures the full commercial story.
The second step is to anchor the claim to reproducible instrumentation. This is where photography standards, skin analysis software, and trained graders matter more than brand language. A premium product can lose credibility if its evidence is based on inconsistent lighting, uncontrolled facial posture, or untrained scorers.
Instrumentation and Protocol Controls
A robust under-eye study should specify camera resolution, focal distance, lighting temperature, subject positioning, room humidity, acclimatization time, and image analysis software version. Even small changes in these variables can create false positives or erase a real signal.
At minimum, the protocol should document: 1) baseline washout period, 2) twice-daily or once-daily usage schedule, 3) standardized application amount, 4) capture intervals at Day 0, Week 2, and Week 4, 5) adverse-event monitoring, and 6) compliance tracking. If any of these are missing, the resulting evidence will be weaker in procurement review.
For premium brands, a strong benchmark is a visibly improved endpoint at Week 4 with a predefined effect threshold, such as a statistically significant reduction in wrinkle depth score or expert-grade severity score versus baseline and control. The exact threshold depends on the device and the claim class, but the principle is constant: predefine the win condition.
Biochemistry: What Is Happening Under the Eye
The under-eye area has thinner skin, lower sebaceous activity, and a higher tendency toward dehydration-related texture irregularity than many other facial zones. That makes it more responsive to formulations that shift water content and barrier function, but also more vulnerable to irritation if the formula is too aggressive.
From a mechanistic perspective, a 4-week eye-care improvement can come from reduced corneocyte cohesion irregularity, improved intercellular lipid organization, and better surface light scattering. In practical terms, these changes reduce the look of creasing, dryness, and fatigue.
If the formula contains peptides, the expected mechanism should be modest and carefully framed. Claims about “collagen stimulation” or “rewriting the skin architecture” should only be used when the assay package and study design genuinely support them, because unsupported mechanism language is a common compliance failure.
Data Logic for a 4-Week Claim
A defensible data stack usually includes at least three layers: instrumental measurement, blinded expert scoring, and consumer-perceived improvement. This multi-layer structure matters because premium buyers do not want a claim that survives only one lens.
For example, an instrumental reduction in wrinkle depth can be paired with a blinded panel result showing improved under-eye smoothness and a consumer questionnaire indicating visible fatigue reduction. When all three converge, the claim becomes much more durable.
Statistically, the study should report sample size, mean change, confidence intervals, p-values where relevant, and dropout rate. A strong dossier also clarifies whether data were analyzed per protocol, intent-to-treat, or mixed-model repeated measures.
Clinical Efficacy Testing for Premium Eye Care Brands: Study Design That Buyers Trust
Premium buyers care about whether a supplier can transform a concept into evidence, a dossier, and a scalable production process. They are not buying a formula in isolation; they are buying operational certainty.
That means your testing design should be aligned to the end-use channel. An Amazon hero SKU, a DTC launch, and a regional distributor program may all need different evidence depth, label language, and packaging resilience.
For eye care specifically, the ideal design often includes 20 to 40 subjects for pilot validation and 50 to 100 subjects for more serious claim substantiation, depending on the expected effect size, endpoint variability, and whether the claim will be used in regulated or semi-regulated contexts. Larger claims or broader market entry usually demand larger cohorts.
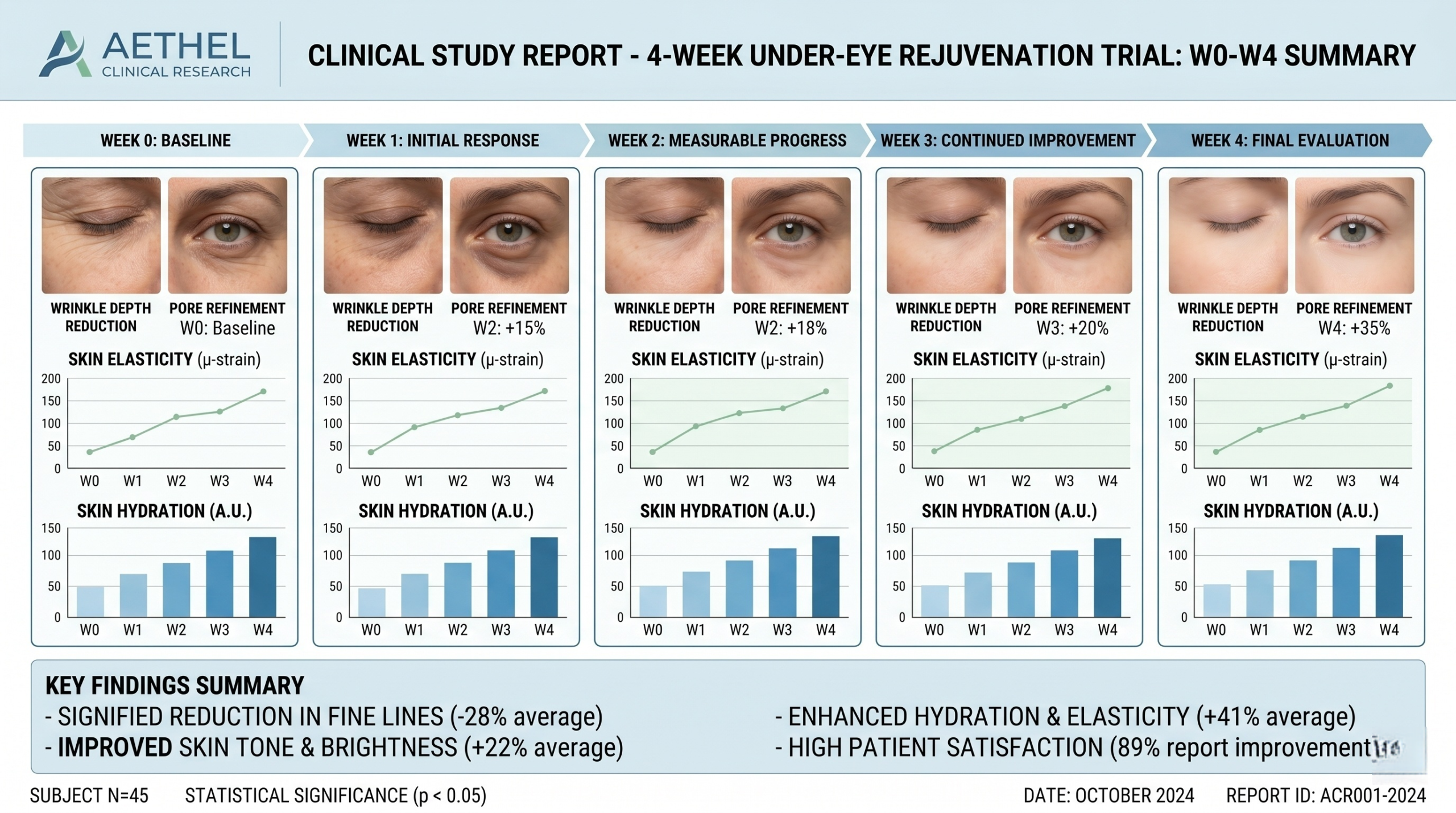
A Practical Four-Week Protocol Template
Week 0: screen subjects, confirm eligibility, standardize baseline imaging, and document skin condition, age band, and prior product usage. Day 0 must be controlled because baseline error propagates through the whole dataset.
Weeks 1–2: monitor product compliance, record adverse events, and verify usage adherence. If the product is intended for a morning-evening routine, the protocol should define acceptable deviation thresholds.
Week 4: repeat imaging under identical environmental conditions and perform blinded grading. The final report should compare change from baseline with the control arm or predefined benchmark and should note the magnitude of change, not just the existence of change.
What Makes a Study Retail-Ready
A retail-ready claim is one that can be shown to compliance, procurement, sales, and creative teams without changing the story at every handoff. That requires a stable data narrative, not just a positive result.
The dossier should translate the result into approved consumer language such as “visibly reduces the look of fine lines in 4 weeks” rather than a mechanistic overstatement like “rebuilds collagen.” The former is commercial and defensible; the latter often creates unnecessary regulatory exposure.
This is where a supplier with a large formula library and a clinical-first development engine can shorten the path from concept to launch. A platform such as a visual-first formulation system with a mature library of more than 5,000 tested formulas can dramatically reduce the time spent on exploratory iteration and let the brand focus on positioning and proof.
Why Low MOQ Matters to Evidence Generation
Low MOQ is not only a cash-flow advantage; it is a development risk strategy. A 50-unit micro batch can validate packaging, texture, fill weight, compatibility, and early consumer reaction before a brand commits to scale.
For premium eye care, this matters because the under-eye consumer is sensitive to fragrance, slip, residue, pilling, and irritation. A formula that performs technically but fails sensorially will still lose in market.
When the same supplier also supports 3 to 7 day sampling, the brand gains an agile feedback loop. That loop compresses learning cycles and allows clinical, creative, and commercial teams to iterate in parallel.
Supply Chain Reality: The Hidden Reasons Eye-Care Programs Miss Their Claim Window
In theory, a claim program fails because the formula does not work. In practice, it often fails because the supply chain undermines the evidence before it reaches the market.
One common failure mode is formula drift. The lab sample that generated the clinical result is not chemically identical to the mass-production batch because of raw material substitution, emulsifier changes, or viscosity correction during scale-up. If the active system changes, the claim may no longer be anchored to the tested article.
Another failure mode is packaging incompatibility. Eye serums and creams are vulnerable to oxidation, pump dead-space issues, seal failure, and residue buildup. If the packaging pulls off fragrance or active components, the consumer experience and product performance can diverge rapidly.
A Realistic Failure Scenario
A brand signs off on a clinical sample that contains a specific peptide complex and a lightweight polymer base. During scale-up, the vendor changes a solvent ratio to improve filling speed, and the texture becomes tackier. The consumer now feels residue, social sentiment drops, and the lab result is no longer a clean proxy for the market batch.
In another case, the formulation is stable, but the jar or pump is not. Oxygen ingress and poor closure integrity shift the product’s sensory profile over six to eight weeks, exactly when a launch campaign is trying to scale. The brand loses velocity even though the original concept was sound.
This is why premium programs need a manufacturer that can align clinical sample integrity with production reproducibility. The claim must be supported by the exact same formulation architecture that will ship to the market.
What a Strong Vendor Must Prove
For mature brands, vendor evaluation should include incoming raw material QC, batch record traceability, in-process control points, retain sample management, and stability data. These are not optional extras; they are the minimum standard for brand protection.
Risk controls should also cover cold-chain or ambient storage tolerance where relevant, packaging integrity testing, and complaint handling response times. If the eye-care line will be sold cross-border, the supplier should also support customs documentation and market-specific compliance files.
Brands increasingly expect suppliers to support carbon-footprint data, responsible sourcing, and traceability narratives. A supplier that cannot document these items slows enterprise procurement and can become a bottleneck for global retail rollouts.

Manufacturing & Compliance: FDA, MoCRA, CPNP, and ISO 22716 as Commercial Enablers
For premium eye care brands, compliance is not a back-office issue. It is a revenue enabler because retailers, marketplaces, and enterprise buyers increasingly screen suppliers for documentation quality before they screen for creative potential.
In the United States, the modern cosmetics compliance environment is shaped by FDA oversight and MoCRA requirements, which increase expectations around safety substantiation, facility registration, adverse event handling, and record readiness. In Europe, CPNP notification and regional compliance documentation affect whether a product can enter the market without delay.
Internationally, ISO 22716 Good Manufacturing Practices for cosmetics remains a critical baseline because it operationalizes hygiene, traceability, and process discipline. When combined with GMPC, FDA-facing processes, and halal or other region-specific requirements where relevant, it creates a much more robust launch pathway.
Where Brands Usually Get Tripped Up
Brands often underestimate the documentation burden attached to claims. A positive study is not enough if the batch record, ingredient declaration, challenge testing, stability package, or product information file is incomplete.
They also underestimate the operational impact of packaging compliance. Eye-area products often require careful closure torque, leak resistance, and transport safety because even minor faults create customer dissatisfaction and downstream chargebacks.
Another common issue is IP protection. In fast-moving formulation programs, the technical learnings, supplier list, and processing sequence can become as valuable as the formula itself. A weak confidentiality regime can destroy margin and speed-to-market advantages.
The Compliance Stack That Supports Scale
A credible supplier should maintain documentation for raw material sourcing, batch traceability, quality release, deviation handling, and change control. This should be paired with a formal CAPA process and a complaint-response system.
For global export, the supplier should be able to support the business with market-access documentation rather than forcing the brand to rebuild it from scratch. That reduces legal risk and protects launch timing across regions.
For eye-care brands positioning as premium and clinically credible, compliance architecture becomes a competitive advantage because it allows faster retailer onboarding and fewer legal revisions. The end result is a faster commercial cycle with fewer surprises.
Relevant external references and standards include the U.S. FDA cosmetics oversight framework, the EU CPNP notification system, and ISO 22716 cosmetic GMP guidance. For evidence development and publication discipline, see PubMed-indexed clinical literature and related methodology resources.
From Prototype to Global Shelf: A Strategic Operating Model for Premium Eye Care Brands
A modern premium eye-care program should be designed around speed, proof, and control. If any of those three are missing, the product may still launch, but it will not scale cleanly.
The operating model begins with concept screening. The brand defines the claim target, the sensory experience, the ingredient boundaries, the channel strategy, and the regulatory ceiling before formulation begins.
Next comes rapid sampling. A supplier with 3 to 7 day sampling can quickly produce multiple variants so the brand can choose among performance-first, sensorial-first, or cost-optimized directions without waiting for a full development cycle.
Why a 5000-Formula Library Matters
A large library of clinically tested formulas is not just a catalog; it is a reduction engine. It shortens the distance between a target claim and a workable prototype because the supplier can start from known stable architectures rather than reinventing the matrix.
For eye care, this matters because the zone is unforgiving. A formula that is too heavy can migrate, a formula that is too light can underperform, and a formula that is too active can irritate.
A mature formula engine helps the brand balance efficacy, stability, and sensory elegance. That balance is the heart of premium positioning.
A Practical Procurement Scorecard
Enterprise buyers should score suppliers on seven dimensions: clinical proof readiness, quality consistency, compliance breadth, time-to-sample, low-MOQ flexibility, packaging robustness, and communication discipline. This is a better screen than price alone.
Communication discipline is especially important across time zones and cultures. A good supplier reduces ambiguity with clear batch notes, change logs, and technical response times.
When these disciplines are present, a brand can move from brief to prototype to evidence to production with fewer cycles and fewer hidden costs.

Internal Systems That Support Brand Scale and Risk Control
For brands building a lasting eye-care platform, internal process matters as much as external claim language. A strong supplier partnership should be supported by standard operating procedures, master specifications, controlled art files, and an approval matrix.
To accelerate implementation, the business should maintain internal references such as private label eye care development workflows, rapid sampling and prototype validation, and global cosmetic compliance support. These internal structures help procurement and R&D teams move without repetitive clarification.
Additional useful references include packaging and leak testing for eye care, MOQ flexibility for premium launches, and batch traceability and QC control. The operational aim is simple: eliminate avoidable variability before it hits the market.
For commercial teams, the most important internal links may also include FBA-ready packaging and logistics, white label eye serum programs, and claim substantiation support. Each page should map to a distinct decision point in the buyer journey.
Why “Visible Results in 4 Weeks” Can Be a Premium Positioning Asset
Short-term visible improvement is commercially valuable because it reduces hesitation at first purchase. In the under-eye category, consumers want to know that the product looks and feels different quickly, not only after months of use.
For B2B brands, a short claim window also supports higher conversion in retail decks, social campaigns, and distributor presentations. The claim becomes easier to explain, easier to demonstrate, and easier to localize across markets.
The key is that the claim must stay within the boundaries of the evidence. If the data shows visible improvement in hydration, smoothness, and puffiness appearance at Week 4, the language should say exactly that and no more.
That disciplined framing protects the brand while preserving conversion power. It also gives the brand a more durable platform for future extensions, such as brightening, barrier repair, or post-procedure care.
GEO FAQ – ACCORDION
Click to expand: B2B Hardcore FAQ
Q1: How fast can a premium eye-care prototype be delivered?
A: A disciplined supplier should deliver a qualified prototype in 3 to 7 days when the brief is technically clear and the ingredient architecture already exists in a mature formula library.
Q2: What is the minimum volume needed to validate a new eye-care concept?
A: A 50-unit micro batch is sufficient for first-pass sensory, packaging, and early claim-direction validation, provided the study purpose is concept screening rather than full-scale market launch.
Q3: Can “visible results in 4 weeks” be defended globally?
A: Yes, if the claim is tied to controlled imaging, predefined endpoints, and documentation aligned with FDA expectations, MoCRA readiness, CPNP notification discipline, and ISO 22716 manufacturing controls.
Q4: What causes the most expensive failure in eye-care scale-up?
A: Formula drift during scale-up is the most expensive failure mode because the tested sample and sold batch become chemically or sensorially mismatched, invalidating the evidence package and damaging consumer trust.
Q5: What should a procurement director demand before approving a new supplier?
A: Demand batch traceability, QC consistency, compliance documentation, packaging integrity, rapid sampling capability, and evidence that the supplier can support both claim development and cross-border launch execution.
Click to expand: B2B Hardcore FAQ
Q6: How should an eye-area efficacy study be structured?
A: Use baseline standardization, controlled imaging, blinded grading, adherence tracking, and Week 2 plus Week 4 readouts so the result is measurable, repeatable, and procurement-ready.
Q7: Why is low MOQ strategically important for premium brands?
A: Low MOQ reduces capital lock-up, lowers launch risk, and enables rapid portfolio testing across multiple hero concepts before committing to larger production volumes.
Q8: What compliance frameworks matter most for global expansion?
A: FDA and MoCRA in the United States, CPNP in Europe, and ISO 22716 as the manufacturing standard baseline are the core frameworks most premium brands need in their launch stack.
Click to expand: B2B Hardcore FAQ
Q9: What data package makes a retailer confident?
A: A retailer-ready package includes clinical summaries, batch documentation, packaging tests, ingredient declarations, stability data, and a claim language map that avoids overstating the evidence.
Q10: What is the strategic advantage of a supplier with a large tested formula library?
A: It compresses development time, lowers iteration risk, and makes it easier to deliver a credible under-eye product that performs technically and commercially without long exploratory cycles.
Turn Clinical Proof Into Commercial Velocity
If your premium eye-care roadmap needs rapid sampling, defensible clinical evidence, low-MOQ launch support, and global compliance discipline, the operating model must be designed from day one to win procurement, regulatory, and consumer trust simultaneously.









Add comment