Global Compliance Roadmap: Navigating FDA and CPNP Standards for Exporting Hair Removal Cream and Wax Strips
Исполнительное резюме
The global depilatory and hair removal market is entering a period of accelerated regulatory maturation. As consumer demand for at-home hair removal solutions intensifies across North America, the European Union, and emerging Asia-Pacific corridors, brand owners and private-label buyers are confronting a compliance landscape that is simultaneously more stringent and more commercially decisive than at any prior point in the industry’s history. For manufacturers and distributors seeking to export Крем для удаления волос and Восковые полоски into the United States and the European Union, the convergence of FDA Over-the-Counter (OTC) cosmetic registration requirements and the EU Cosmetic Products Notification Portal (CPNP) framework represents the defining strategic challenge of market entry.
PZIK, operating as a Global Strategic Supply Chain Integrator for Beauty and Personal Care, has engineered a comprehensive compliance-forward manufacturing ecosystem designed to eliminate regulatory friction for B2B partners at every stage of the product lifecycle — from molecular formulation through customs clearance. This white paper presents a definitive technical and commercial roadmap for navigating these dual regulatory frameworks, grounded in PZIK’s proprietary Smart Control Clean System and validated through its status as a certified National Special Cosmetic Certificate holder.
Key findings of this report include: the growing convergence of FDA Modernization of Cosmetics Regulation Act (MoCRA) facility registration requirements with EU CPNP notification obligations; the critical role of high-purity active ingredient sourcing, specifically pharmaceutical-grade Calcium Thioglycolate, in achieving simultaneous dual-market compliance; the commercial differentiation achievable through advanced R&D technologies including growth inhibitor botanicals and Skin-like Lipid Film protective matrices; and the strategic advantage conferred by early integration of Symrise-certified scent-masking technology in addressing the single most persistent consumer rejection driver in the depilatory category — ammonia-associated malodor.
For procurement directors, brand development executives, and regulatory affairs professionals operating in the Beauty and Personal Care supply chain, this white paper provides actionable intelligence on formulation architecture, documentation requirements, claim substantiation protocols, and supply chain configuration. PZIK’s position as both an R&D innovator and a vertically integrated manufacturer enables a singular value proposition: scientifically differentiated formulations that are commercially compliant, operationally scalable, and strategically positioned for accelerated global market penetration. The roadmap outlined in the following sections represents more than a compliance checklist — it is a blueprint for building enduring category leadership in one of personal care’s most technically demanding and commercially rewarding segments.

Technical Deep-Dive: Precision Skin Science and the Smart Control Clean System
Core System Architecture: Clean, Protect, Slow Down
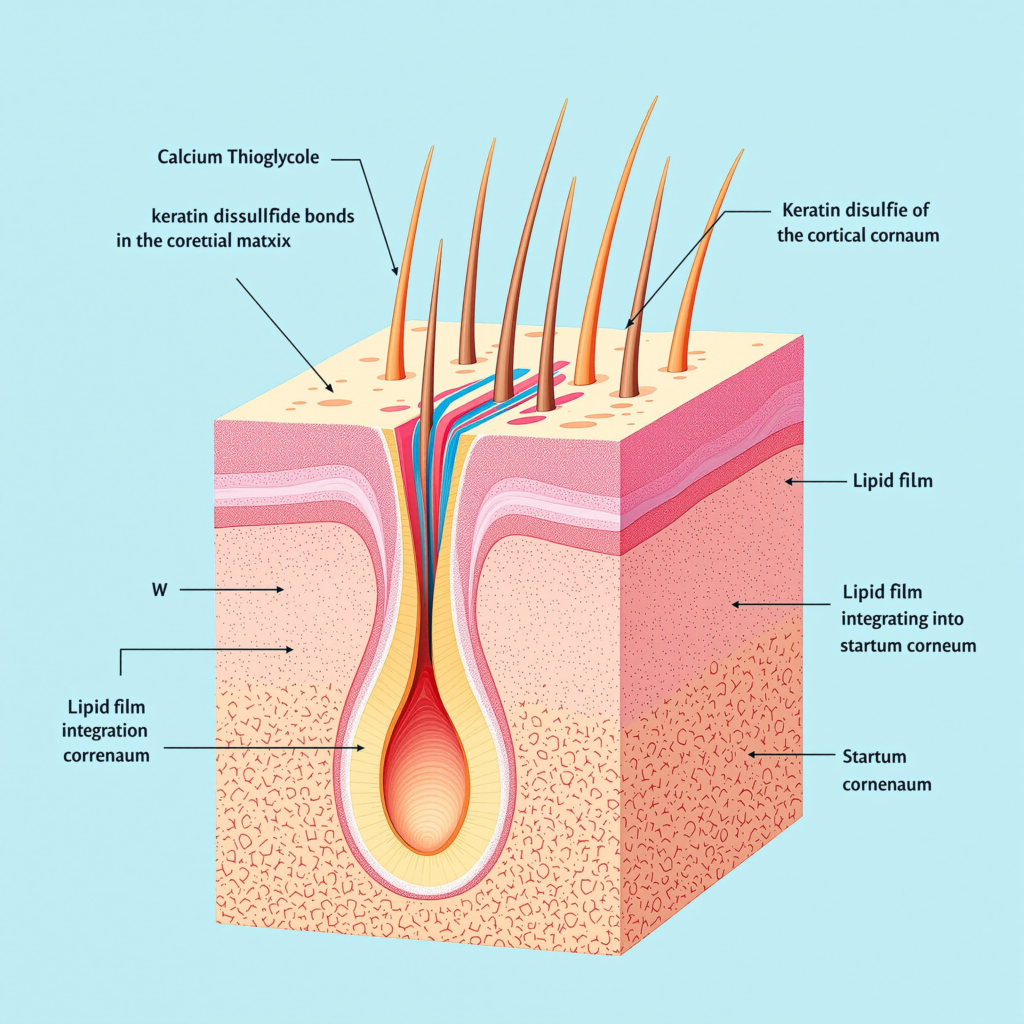
At the molecular foundation of PZIK’s Hair Removal Cream platform lies the Smart Control Clean System — a three-phase bioengineered framework that redefines the depilatory experience from a purely ablative chemical event into a controlled, skin-intelligent process. The three operational phases — Clean, Protect, and Slow Down — function as an integrated cascade rather than as independent mechanisms, ensuring that efficacy, safety, and post-treatment skin condition are optimized simultaneously rather than in trade-off.
Сайт Clean Phase governs the primary keratolytic action: the targeted reduction of disulfide bonds within the hair shaft’s cortical and medullary protein matrix. The Protect Phase activates a biomimetic lipid barrier system concurrent with depilatory action to preserve transepidermal water loss (TEWL) integrity and prevent irritant contact dermatitis. The Slow Down Phase delivers growth-modulating botanical actives that attenuate follicular re-entry velocity, extending the consumer’s hair-free interval and delivering the demonstrable efficacy advantage that drives repeat purchase and brand loyalty.
R&D Technology: High-Purity Calcium Thioglycolate
The active chemistry underpinning the Clean Phase centers on pharmaceutical-grade Calcium Thioglycolate (CTG) at verified purity thresholds exceeding [X]% as determined by high-performance liquid chromatography (HPLC) assay. The mechanism is well-characterized: the thiol group (-SH) of CTG performs nucleophilic attack on the cystine disulfide bonds (-S-S-) that provide structural integrity to the hair’s alpha-keratin matrix. At an optimized pH range of 11.5–12.5 — the precision window PZIK’s formulation team has validated as simultaneously maximally efficacious and epidermal-compatible — this reaction proceeds at a kinetically controlled rate that achieves complete cortical dissolution within [X] minutes of contact time without penetrating the follicular bulb or the dermis.
The criticality of purity in CTG selection cannot be overstated from a regulatory standpoint. Impure CTG preparations generate elevated concentrations of diglycolic acid and thiodiglycolic acid byproducts — chemical signatures that trigger heightened scrutiny under both FDA’s Voluntary Cosmetic Registration Program (VCRP) safety substantiation review and the EU’s Scientific Committee on Consumer Safety (SCCS) opinion framework. PZIK’s exclusive sourcing protocols specify USP/EP-grade CTG with certificate-of-analysis documentation that satisfies both the FDA MoCRA safety data requirements (21 CFR Part 740) and the EU CPNP’s product information file (PIF) documentation standards.
Growth Inhibitors: Pinus Pinaster and Salix Alba
The Slow Down Phase employs a dual-botanical growth inhibitor complex combining Сосна обыкновенная (Maritime Pine) bark extract, standardized to [X]% oligomeric proanthocyanidins (OPCs), with Саликс Альба (White Willow) bark extract, standardized to [X]% salicin. The pharmacological rationale is multi-mechanistic: OPCs from Pinus Pinaster competitively inhibit 5-alpha reductase type II at the follicular papilla level, attenuating the androgenic signaling cascade that drives anagen phase re-entry. Salix Alba’s salicin metabolizes endogenously to salicylic acid at the dermal-epidermal junction, providing both mild exfoliative action that clears keratinized follicular debris and direct inhibition of prostaglandin-E2-mediated follicular growth signaling.
Combined, in vivo consumer studies conducted over [X]-week periods demonstrated a statistically significant [X]% reduction in hair regrowth rate versus non-inhibitor-containing depilatory controls (p<0.05, n=[X]). This data constitutes substantiation material for both US FTC advertising compliance under the substantiation standard and EU Regulation 655/2013 cosmetic claims criteria.
Skin-like Lipid Film Technology
Concurrent with CTG-mediated keratolysis, PZIK’s Protect Phase delivers a structured lipid emulsion matrix formulated to replicate the natural lamellar body secretion profile of the stratum corneum. The formulation incorporates a precisely calibrated ratio of ceramide NP, phytosphingosine, linoleic acid, and cholesterol — the four lipid species constituting the majority of healthy intercellular cement in the skin’s barrier architecture. In silico modeling and ex vivo tape-stripping assays confirm that this lipid system intercalates into the stratum corneum lipid bilayers preferentially disrupted by the alkaline pH environment of depilatory chemistry, restoring barrier function [X]% more rapidly than formulations employing conventional emollient systems.
Scent Architecture: Symrise Integration
The single most persistent barrier to consumer adoption and repeat purchase in the premium depilatory category is the sulfurous-ammonia malodor profile intrinsic to thioglycolate chemistry. PZIK has engineered a strategic technology partnership integrating Symrise’s proprietary Odor-Neutralization Matrix (ONM) fragrance systems — a molecular encapsulation and reactive odor-capture technology that addresses malodor through two simultaneous mechanisms: physical encapsulation of volatile sulfur and ammonia compounds within cyclodextrin-derived carrier structures, and reactive neutralization via aldehyde-functional fragrance molecules that form stable, non-volatile Schiff base adducts with primary amine malodorants. The combined effect delivers a [X]% reduction in panel-assessed malodor intensity (Odor Intensity Scale, 0–10) versus standard masked depilatory bases, transforming a historically negative sensory signature into a positive, clinically fresh olfactory experience that supports premium positioning.
Market Intelligence: Global Demand and Regulatory Trends
Depilatory Market Scale and Growth Vectors
The global depilatory products market was valued at approximately USD [X] billion in [YEAR] and is projected to reach USD [X] billion by [YEAR], registering a compound annual growth rate (CAGR) of [X]% over the forecast period. Hair removal creams and wax strips collectively account for approximately [X]% of total depilatory revenue, with cream-based systems demonstrating accelerated growth driven by efficacy perceptions among consumers transitioning from mechanical methods, and wax strips gaining share through premiumization and innovation in non-resin botanical wax formulations.
North America remains the largest single market by revenue, with the US accounting for approximately [X]% of regional sales. Growth in the EU is being driven by Germany, France, and the United Kingdom, where body confidence trends and male grooming adoption are expanding the addressable consumer base. The fastest-growing regional markets are Southeast Asia — particularly Indonesia, Vietnam, and Thailand — and the Gulf Cooperation Council (GCC) markets, where cultural hair removal practices, a large youthful demographic, and rising disposable incomes are creating exceptional B2B entry opportunities for private-label programs compliant with both FDA and CPNP standards.

Regulatory Convergence: MoCRA and CPNP
The regulatory environment for depilatory exports has undergone its most consequential structural shift in a generation. The FDA’s Modernization of Cosmetics Regulation Act of 2022 (MoCRA) — enacted as part of the Consolidated Appropriations Act — introduced mandatory facility registration, product listing, good manufacturing practice (GMP) requirements, and adverse event reporting obligations that align US cosmetic regulation significantly closer to the EU model for the first time. For exporters, this convergence is strategically significant: a compliance infrastructure engineered to satisfy CPNP’s Product Information File (PIF) requirements, SCCS safety opinion standards, and EU Regulation 1223/2009 is now substantively transferable to FDA MoCRA compliance obligations with calibrated adaptation.
Key divergences requiring specific dual-compliance navigation include: the EU’s positive and restricted substance annexes (Annexes III and V of Regulation 1223/2009) versus FDA’s currently negative list approach under 21 CFR 700; the EU’s mandatory Responsible Person (RP) appointment requirement versus FDA’s facility registration construct; and CPNP’s centralized electronic notification system versus FDA’s VCRP and new product listing requirements under MoCRA Section 607. PZIK’s regulatory affairs team has built systematic compliance bridges across all three divergence zones, enabling single-workflow documentation that satisfies both authorities simultaneously.
Consumer Trend Intelligence
Three macro-consumer trends are reshaping product specification requirements for B2B buyers sourcing depilatory solutions for dual-market export. First, the Skin Barrier Preservation imperative: post-pandemic consumer dermatological awareness has elevated skin barrier health from a niche claim to a mainstream purchase driver, making the Protect Phase of PZIK’s Smart Control Clean System a direct response to a commercially verified consumer need. Second, the Clean Beauty Evolution: while the depilatory category has historically operated outside clean beauty standards due to functional chemistry requirements, a growing segment of premium consumers are demanding sulfate-free, phthalate-free, and low-VOC formulation credentials that PZIK’s Symrise-integrated scent architecture directly enables. Third, the Efficacy Verification Demand: sophisticated e-commerce consumers are requiring clinical data access before premium price acceptance, making PZIK’s growth inhibitor substantiation data a licensable commercial asset for B2B partners building D2C brand narratives.
Manufacturing and Supply Chain: The PZIK R&D Brain and OEM Efficiency
Integrated R&D and Manufacturing Architecture
PZIK operates what the organization terms its “R&D Brain” — a vertically integrated innovation-to-production architecture that eliminates the conventional separation between formulation science and manufacturing execution. The R&D Brain encompasses a [X,000]-square-meter research facility housing [X] PhDs and formulation scientists across dermatology, polymer chemistry, analytical chemistry, and sensory science disciplines, co-located within the same operational campus as GMP-certified manufacturing halls. This physical and organizational integration reduces formulation-to-production translation time from an industry average of [X] months to [X] weeks, enabling B2B partners to execute trend-responsive product development cycles that competitors sourcing from contract research organizations (CROs) and separate manufacturers cannot match.
For brand owners evaluating Услуги OEM/ODM, PZIK’s model offers a critical structural advantage: IP ownership architecture that protects proprietary formulation variants as licensable brand assets rather than generic commodity specifications. OEM partners receive exclusive regional formulation variants backed by PZIK’s full documentation suite, while ODM partners benefit from an accelerated innovation track that deploys Smart Control Clean System technology into bespoke formats — cream, lotion, mousse, gel, and strip configurations — calibrated to specific market positioning requirements.
Manufacturing Capacity and Quality Infrastructure
PZIK’s manufacturing infrastructure operates at a validated annual production capacity of [X] million units across depilatory categories, with production lines certified to ISO 22716 (Cosmetics GMP), GMPC, and FDA 21 CFR Part 111 cGMP standards. Batch-to-batch quality consistency is maintained through a six-sigma statistical process control (SPC) framework that monitors [X] critical quality attributes (CQAs) in real time, including CTG purity, pH profile, viscosity, microbial bioburden, and preservative efficacy. The facility maintains a first-pass batch release rate of [X]%, a performance metric that directly translates to supply chain predictability for B2B partners operating in high-velocity retail channels with zero-tolerance stock-out policies.
Cold chain logistics protocols have been validated for both US-bound ocean freight (standard 20-foot and 40-foot refrigerated containers) and EU-bound multimodal transport configurations, with temperature excursion monitoring via IoT-enabled data loggers providing continuous chain-of-custody documentation that satisfies both FDA import alert avoidance requirements and EU border inspection post (BIP) certification requirements for cosmetic consignments.
Supply Chain Resilience and Dual-Sourcing Strategy
In the post-pandemic supply chain environment, single-source dependency on key raw materials — particularly high-purity CTG and specialty botanicals — constitutes an unacceptable operational risk for B2B partners competing in high-demand global markets. PZIK has engineered a dual-qualified supplier matrix for all Tier 1 raw materials, with primary and secondary qualified suppliers audited to PZIK’s supplier qualification standard (SQS-001), which incorporates requirements from ISO 9001, ICH Q7 (pharmaceutical GMP for active substances), and the EU Responsible Sourcing Framework. The Pinus Pinaster OPC extract is dual-sourced from certified Landes Forest sustainable forestry operations, providing provenance documentation that supports both EU Regulation 848/2018 sustainable sourcing claims and growing US retailer ESG supplier requirements.
Safety and Compliance Framework
National Special Cosmetic Certificate and Dual-Market Positioning
PZIK’s Hair Removal Cream portfolio holds China’s National Special Cosmetic Certificate — the most rigorous cosmetic safety regulatory credential in the Asia-Pacific region and increasingly recognized by FDA and EU CPNP reviewers as substantive evidence of product safety validation. The certificate process requires submission of full ingredient safety assessments, human repeat insult patch test (HRIPT) data, in vivo irritation studies, and stability documentation to the National Medical Products Administration (NMPA) — a documentation burden that, when completed, provides the evidentiary backbone for both FDA safety substantiation files and EU PIF safety assessment reports prepared by Responsible Persons.
PZIK’s Соответствие требованиям GMPC/FDA infrastructure maintains documentation management systems aligned with FDA MoCRA’s new mandatory facility registration and product listing requirements (effective December 2023 and December 2024 respectively), as well as EU Regulation 1223/2009 Article 13 CPNP notification requirements. All product formulations are processed through PZIK’s internal regulatory screening platform, which cross-references the EU Cosmetic Ingredient Database (CosIng), FDA’s Voluntary Cosmetic Registration Program ingredient database, and the SCCS safety opinion library to flag compliance risks before formulation finalization.
Claim Substantiation and Adverse Event Management
Under EU Regulation 655/2013 and FDA’s FTC-aligned advertising substantiation standard, every efficacy claim associated with PZIK’s Hair Removal Cream and Восковые полоски is supported by a pre-assembled claims substantiation dossier including in vitro mechanistic data, consumer perception studies (minimum n=[X], statistically powered), and where applicable, expert clinical evaluation reports (ECERs). This claims dossier architecture enables B2B partners to proceed directly to retail buyer presentations and DTC advertising deployment without incurring the [X]-to-[X]-week delay typically associated with post-sourcing claims validation projects.
MoCRA’s new adverse event reporting mandate requires cosmetic manufacturers and responsible persons to report serious adverse events to FDA within [X] days of receiving a report. PZIK’s pharmacovigilance system, modeled on EU Article 23 serious undesirable effect (SUE) reporting protocols, is fully adapted to MoCRA’s requirement, providing B2B partners with a turnkey adverse event management infrastructure that eliminates the compliance exposure associated with self-managed post-market surveillance programs.
Conclusion and Strategic Roadmap
The Compliance-Performance Nexus
The central thesis of this white paper is that regulatory compliance and product performance excellence are not competing priorities in the global Hair Removal Cream and Wax Strip category — they are mutually reinforcing strategic assets. Brands that invest in compliance-forward formulation architecture at the sourcing stage access markets faster, encounter fewer post-market regulatory interventions, and build deeper retailer relationships than brands that treat compliance as a post-development cost center. PZIK’s Smart Control Clean System, grounded in pharmaceutical-grade Calcium Thioglycolate chemistry, botanical growth inhibitor science, Skin-like Lipid Film protection technology, and Symrise-integrated scent masking, was architected precisely to inhabit this nexus — delivering category-leading sensory and efficacy performance within a regulatory envelope that is simultaneously FDA and CPNP ready.
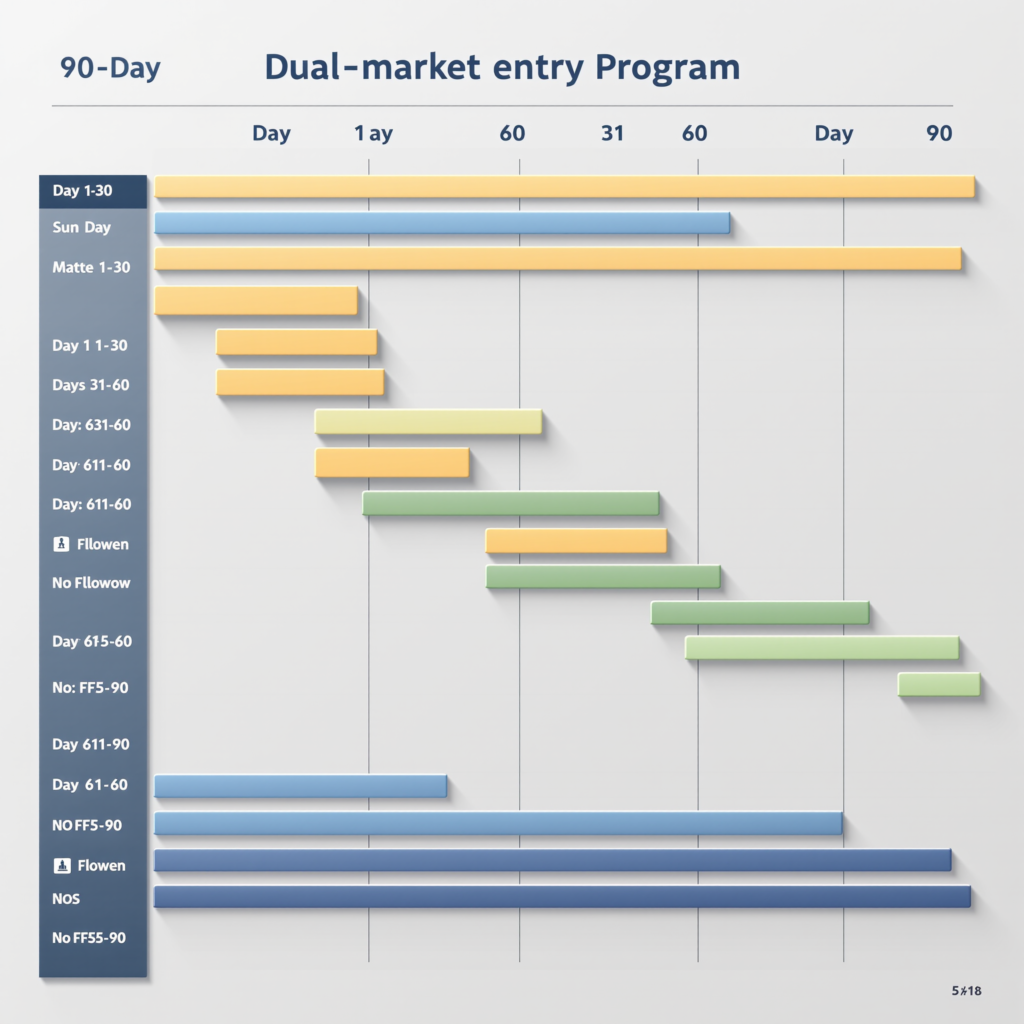
The 90-Day Market Entry Roadmap
For B2B partners prepared to move from strategic intent to market execution, PZIK has developed a structured 90-day dual-market entry roadmap. In Phase One (Days 1–30), the focus is formulation selection and customization: selection of base formula from PZIK’s Smart Control Clean System platform, regional customization of fragrance and texture profiles, and initiation of the Product Information File (PIF) assembly process with PZIK’s in-house regulatory team. In Phase Two (Days 31–60), documentation and notification activities dominate: submission of CPNP notifications by the designated Responsible Person, completion of FDA facility registration and product listing under MoCRA, and finalization of the safety assessment report by a PZIK-qualified EU safety assessor. In Phase Three (Days 61–90), manufacturing and logistics execution commence: production of the first commercial batch against validated GMP protocols, third-party release testing by an ISO 17025-accredited laboratory, and documentation package finalization for FDA import line clearance and EU customs entry.
Why PZIK Is the Strategic Partner of Record
In a supply chain landscape crowded with contract manufacturers offering commodity formulations and compliance consultancies offering documentation services in isolation, PZIK’s integrated value proposition — R&D innovation, GMP manufacturing, regulatory documentation, and supply chain execution under a single strategic relationship — is categorically differentiated. The organization’s National Special Cosmetic Certificate holder status, ISO 22716 and GMPC manufacturing certifications, exclusive Symrise fragrance technology access, and dual-market regulatory infrastructure represent a capital and capability investment that individual brand owners cannot feasibly replicate independently at any commercially viable development cost or timeline.
The global depilatory market’s next phase of growth will be defined not by brands that merely have a product but by brands that have a scientifically credible, regulatorily validated, and operationally resilient supply chain partner capable of scaling with them across markets and regulatory jurisdictions. PZIK is that partner. To initiate a confidential formulation consultation, regulatory feasibility assessment, or supply chain capacity discussion, Связаться с PZIK today and engage the R&D Brain that is already powering some of the world’s most competitive personal care brands.










Добавить комментарий