Alpha Arbutin Formulations: 9 EU Compliance Traps Global Brightening Brands Must Avoid
A technical white paper for international beauty brands, supply chain leaders, and sourcing executives building compliant, scalable brightening portfolios.
Market Intelligence: Why Alpha Arbutin Still Wins in Global Brightening, and Why EU Limits Change Everything
Alpha arbutin remains one of the most commercially useful brightening actives because it sits in a rare middle ground: strong enough to support visible tone-evening, but generally more acceptable than hydroquinone in mass-market cosmetics. In practice, its appeal is not only biochemical; it is also logistical, because brands can build maintenance serums, melasma-supporting creams, and post-inflammatory hyperpigmentation products around a familiar INCI with broad consumer trust. For EU-facing programs, however, the formulation brief is not “how much can we add,” but “how do we preserve performance inside the safety envelope?”
The core regulatory challenge is concentration discipline. The European SCCS opinion on alpha-arbutin is the commercial anchor for EU portfolio design, and brands must also respect the operational reality that high-performance claims can collapse if pH, temperature, or contamination are poorly controlled. In other words, the ingredient may be simple on paper, but the system around it is not.
This white paper is written for executive teams who need a supply chain answer, not a cosmetic theory seminar. If you are evaluating an ODM/CMO partner, you should care about hydroquinone impurity control, CPNP readiness, ISO 22716 GMP discipline, batch-to-batch QC stability, and whether your supplier can move from concept to market in 14 days without compromising documentation or release testing.
Technical Deep-Dive – SEO Pillar: Alpha Arbutin Formulations, EU Limits, and the Commercial Math Behind Compliance
The strongest technical argument for alpha arbutin is not that it is the most aggressive depigmenting agent. It is that it can be engineered into a repeatable, scalable, lower-friction brightening platform that works across private label, prestige, dermocosmetic, and marketplace channels. In the peer-reviewed literature, alpha arbutin shows anti-melanogenic activity through tyrosinase-related pathways, and a 2024 randomized split-face study reported that a cream containing alpha-arbutin 5% and kojic acid 2% achieved outcomes comparable to triple combination cream on objective measures while showing lower recurrence and fewer adverse events after discontinuation, based on published data indexed in PubMed Central.
That said, the key commercial lesson is not to blindly copy a 5% academic prototype into the EU. The SCCS position, as summarized in the scientific opinion, supports alpha arbutin up to 2% in face creams and up to 0.5% in body lotions. For global brands, that means the same hero SKU often needs at least two regulatory variants: an EU-compliant version and a higher-dosage version for jurisdictions where the risk assessment, legal position, and product category permit a different dosing strategy.
Alpha Arbutin Mechanism: What Actually Matters for Formulators
Alpha arbutin is a glycosylated hydroquinone derivative. Its functionality depends on enzyme interaction, release behavior, and formulation stability, not marketing language. According to the review literature available via PubMed, arbutin can reduce cellular melanogenesis, influence tyrosinase activity, and contribute antioxidant effects, but the exact magnitude of effect varies with assay type, enzyme origin, substrate conditions, pH, and impurity profile.
The practical implication is straightforward: if your supplier cannot keep the system stable, your ingredient advantage evaporates. Alpha arbutin has been shown in comparative stability work to be more chemically and enzymatically stable than beta-arbutin under certain conditions, but even alpha arbutin can degrade when the formula is too acidic, too hot, or contaminated with hydrolase-producing microbes. A brand that ignores these boundaries is not building a premium serum; it is building a liability.
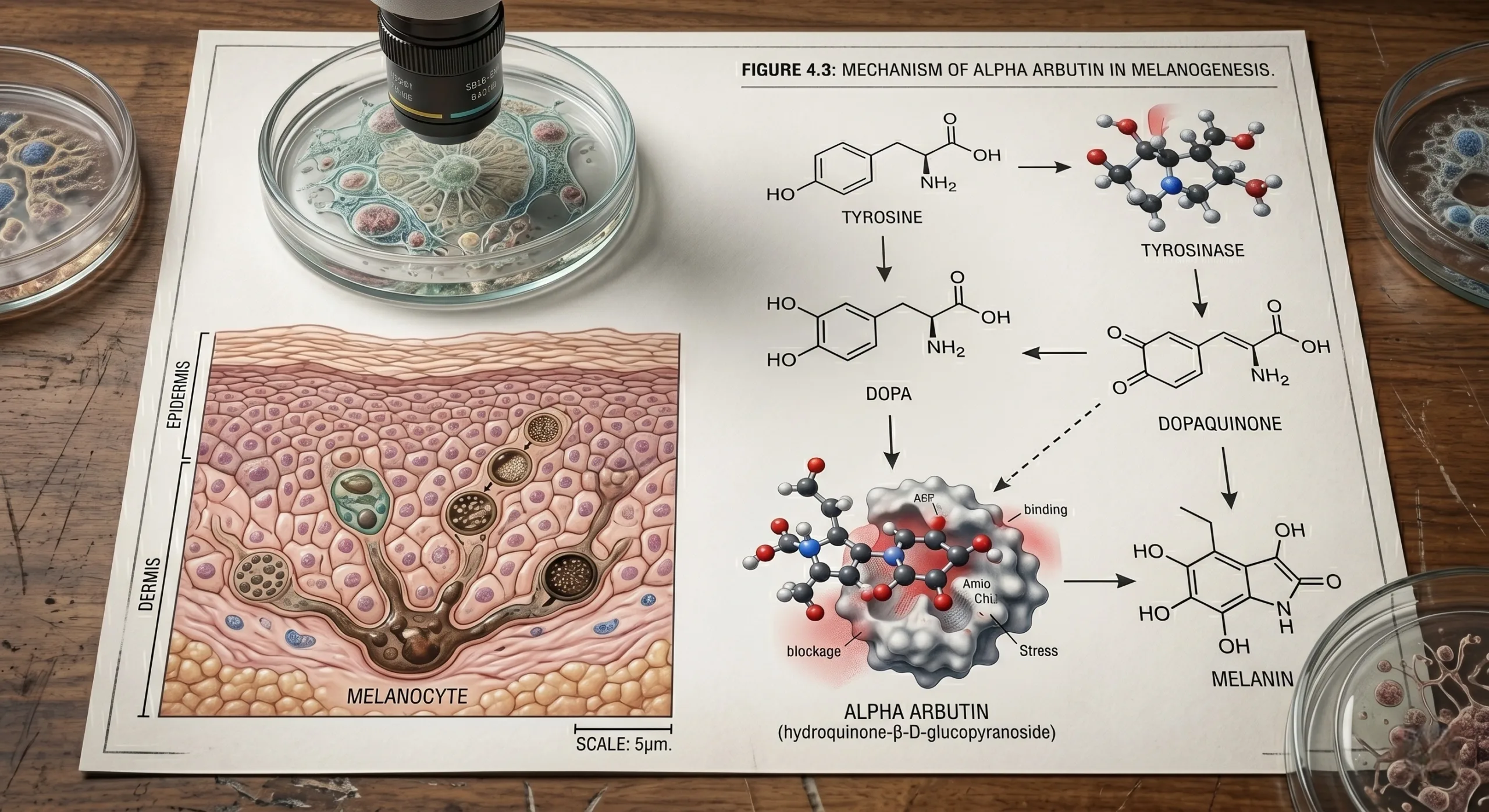
The EU Concentration Logic: Why 2% Is a Strategic Ceiling, Not a Random Number
For EU commercialization, concentration is not a purely technical choice. It is a risk allocation decision that touches safety substantiation, claims defensibility, and downstream retailer acceptance. A 2% face cream ceiling is operationally useful because it gives the formulator a workable efficacy window while keeping the compliance team aligned with the prevailing SCCS safety narrative. Going above that in an EU launch plan increases the burden on toxicological review, product information file quality, and legal review of claims and warning statements.
Brands should understand that “safe” does not mean “indiscriminately scalable.” It means the product can be sold without creating an avoidable compliance gap. If the same formula is sold into ASEAN, the US, and the EU, the smart move is not one universal concentrate. The smart move is a platform formula with jurisdiction-specific actives matrix, preservative system, pH trim, and finished product labeling.
Technical Parameters That Decide Whether the Formula Ships or Fails
In real manufacturing, alpha arbutin performance is strongly affected by measurable process variables. The most important are pH, thermal exposure, metal ion control, preservative compatibility, water activity, packaging barrier integrity, and raw material purity. A formula held at pH 5.5 to 6.5 is typically easier to defend than one drifting toward strong acidity or alkaline stress, because hydrolysis risk rises as the system becomes less controlled.
From a process-control standpoint, the supplier should be able to prove at least these data points: raw material assay above 98% for premium grade alpha arbutin, finished product hydroquinone impurity below 1 ppm where relevant to the safety narrative, prototype turnaround in 3 to 7 days, MOQ starting at 50 pieces for white-label market validation, and scaled pilot runs that preserve viscosity within a narrow tolerance band. A serious partner will also show batch-to-batch pH drift within a controlled range, microbial testing pass rates, and stability retention across 40°C accelerated storage.
What the Literature Says About Efficacy, Without Overstating It
The 2021 review in PMC makes a critical point: alpha arbutin and arbutin have supportive depigmenting evidence, but the exact superiority of one over the other remains debated because assay conditions vary widely. For B2B teams, that means the ingredient should be treated as a supporting brightening active, not as the sole pillar of a melasma portfolio. The best commercial outcomes typically come from combination systems, especially where niacinamide, tranexamic acid, or kojic acid are co-developed to target multiple nodes in the pigmentation pathway.
That strategic approach is supported by the 2024 pilot study on alpha-arbutin 5% and kojic acid 2%, where objective measures suggested performance comparable to triple combination cream, but subjective improvement and recurrence patterns differed. For an international brand, the useful lesson is not “use more alpha arbutin,” but “engineer a product architecture that balances effect, irritation, recurrence, and regulatory resilience.”
Formulation Architecture: Stable, Scalable, and Reviewer-Friendly
A defensible EU brightening formula usually starts with a neutral-to-slightly acidic water phase, chelation support, antioxidant buffering, and a packaging system that minimizes oxygen and light exposure. Airless pumps, opaque tubes, or UV-protective bottles are not cosmetic luxuries; they are stability controls. If the product sits in a clear jar at a 6-month retail window, you are asking the ingredient to survive a bad distribution plan.
Our own formulation philosophy is built around a visual-first, rapid-testing engine with more than 5,000 mature formula patterns, but the commercial value is not “speed” alone. It is the ability to rapidly test a 50-piece white-label concept, identify pH and compatibility failures before production scale-up, and then lock the winning version into a compliance-ready dossier. That is how a brand protects cash flow while still moving at social-commerce speed.

Alpha Arbutin Formulations: The Supply Chain Failure Modes That Cost Brands the Most
Most sourcing problems do not come from the molecule. They come from the ecosystem around the molecule. A brand may approve a promising sample and still fail at launch because the supplier cannot protect the raw material from heat, does not know how to manage hydroquinone trace contamination, or cannot produce a full Product Information File that survives EU scrutiny. The same formula that looks exceptional in a lab can become commercially unsellable if the documentation is weak.
One of the most common pitfalls is the false assumption that alpha arbutin behaves like an ordinary water-soluble cosmetic powder. It does not. It is sensitive to process discipline. Temperature abuse during shipping, prolonged exposure to poor warehouse conditions, and inaccurate pH adjustment can all reduce potency or generate unwanted degradants. If your supplier is unable to explain their incoming material quarantine, retained sample system, and in-process release criteria, your risk profile is already too high.
Case Pattern 1: The Sample That Passed, Then Failed at Scale
We repeatedly see the same pattern in global procurement: a 100 g bench sample performs well, but the 500 kg pilot shows viscosity instability, color shift, or precipitation after one month. The reason is usually not the active ingredient itself. It is the interaction between chelators, humectants, polymeric thickeners, pH adjusters, and the raw material supply chain.
Brands that scale too quickly often ignore container compatibility. An alpha-arbutin serum that is stable in a glass beaker can still fail in an EVOH-limited tube or low-barrier bottle because of oxygen ingress and light transmission. This is why pilot runs must include real packaging, not only lab glass, and why accelerated stability should include at least 40°C, 75% RH where relevant, freeze-thaw cycles, and transport simulation.
Case Pattern 2: The Compliance Gap Hidden Inside a Great Marketing Story
A common commercial trap is the “hero ingredient narrative” that outruns the legal substantiation package. The brand wants to say brightening, spot fading, radiance, and tone evenness. The regulatory file, however, requires evidence-backed language that matches the product category and market rules. In the EU, you need disciplined claims review under the cosmetics framework, while CPNP notification and the PIF must be accurate before launch.
This is where many factories underperform. They can fill bottles, but they cannot help the brand reconcile marketing ambition with claims compliance, INCI accuracy, allergen disclosure, and full traceability. By contrast, a premium partner should provide a launch package that includes batch coding, country-specific labeling guidance, and an ingredient risk register tied to the final formula.
Case Pattern 3: The Microbial Blind Spot
Alpha arbutin can be degraded by microbes under certain conditions, and the literature has documented hydrolysis risk in the presence of human skin bacteria. For brands, this means preservation is not a secondary consideration. It is one of the determinants of active integrity over time.
If the preservative system is weak or the container is repeatedly opened, the product can drift in pH, lose clarity, and eventually suffer a performance collapse that consumers experience as “the serum stopped working.” That is not merely a customer service issue; it is a formulation governance failure. An export-ready factory must be able to articulate challenge testing, preservative efficacy, and microbiological release criteria in plain English.
Manufacturing and Compliance: How to Build an EU-Ready Alpha Arbutin Program Without Rework
A compliant alpha-arbutin platform starts long before filling. It begins with supplier qualification, raw material specification, and a documented understanding of where the active comes from, how it is purified, how it is stored, and what the impurity profile looks like. A B2B buyer should insist on certificate of analysis review, retained sample policy, microbial limits, and stability data before any commercial commitment.
For EU projects, the compliance stack should include ISO 22716 cosmetic GMP alignment, CPNP notification readiness, and a finished product safety assessment. For US expansion, the file should be mapped against MoCRA expectations, including responsible person traceability and serious adverse event processes where applicable. The point is not to overload the project with paperwork; the point is to prevent a last-minute compliance choke point that delays launch by months.
What a Serious Supplier Must Prove on Day One
A real manufacturing partner should be able to evidence at least the following: ISO 22716 implementation, GMPC documentation, batch-level QC records, export packing standards, hydroquinone impurity control, pH monitoring, and a prototype cycle of 3 to 7 days. If they offer 50-piece MOQ white-label validation, they should also explain how retained samples, line clearance, and cross-contamination controls remain valid at micro-batch scale.
Quality cannot be claimed; it must be demonstrated. That includes compatibility between the active and the preservative system, compatibility between the formula and the package, and compatibility between the formula and the destination market’s legal rules. If a supplier cannot walk you through these three compatibilities, they are not a strategic partner; they are a procurement gamble.
The Best-in-Class Launch Stack for Global Brightening Brands
Our recommended architecture for a global alpha-arbutin program is deliberately modular. Use one master formula logic, then create market-specific variants that adjust concentration, fragrance load, claims language, and packaging. That lets the brand preserve its core positioning while avoiding regulatory overshoot in the EU.
For brands with Amazon, DTC, or distributor-driven growth, this modularity matters because channel economics are unforgiving. A lightweight, break-resistant airless package reduces FBA losses, while a stable, low-rework formula reduces returns and customer complaints. In real terms, you are protecting both gross margin and brand reputation.

Practical Formulation Blueprint: What We Recommend for EU and Global Launches
For EU face creams, the rational design target is alpha arbutin at or below 2%, supported by niacinamide, barrier-friendly emollients, a buffered pH system, and a package that resists oxidation. For body lotions, the 0.5% safety framework gives room for maintenance-grade positioning without overpromising. For markets outside the EU, higher concentrations may be possible, but only if supported by local regulatory review and toxicology logic.
From a commercial performance perspective, a serum or cream built around alpha arbutin should avoid unnecessary complexity. Every extra active increases the number of failure points, the number of stability interactions, and the number of claims that must be defended. If the product already has a strong visual transformation story, overloading it can reduce both stability and clarity.
Minimum Technical Specification for a Serious Launch
At minimum, your supplier should be able to support a documented pH target of approximately 5.5 to 6.5, accelerated testing at 40°C, a retained sample protocol, a micro-batch MOQ of 50 units, and a prototype lead time of 3 to 7 days. You should also require airtight control of raw material traceability, with lot-level COA matching and a defined hydroquinone impurity ceiling relevant to the product’s risk assessment.
Brands seeking faster scale should add packaging and logistics metrics to the technical brief. These include drop-test or transit-protection verification, fill-weight consistency, transport shelf-life expectations, and a clear replacement policy for damaged FBA arrivals. A factory that can support both formulation and logistics is reducing your total landed risk, not just your unit cost.
Why Combination Systems Outperform Single-Ingredient Thinking
Alpha arbutin is strongest when it is part of a controlled network. The literature and the market both support that view. Combination systems with niacinamide, tranexamic acid, kojic acid, or supportive botanical brighteners often provide better commercial elasticity than a lone active because they can be tuned for sensitivity, climate, and consumer tolerance.
This matters especially in sensitive-skin segments. The 2024 split-face pilot study found that the alpha-arbutin/kojic acid side had fewer adverse events and lower recurrence than the triple-combination reference side after stopping treatment, which is exactly the sort of product story premium brands want when they position a maintenance brightening SKU. In B2B terms, that means lower churn, fewer complaints, and more repeat purchase potential.
Supply Chain Strategy: From Lab Sample to Global Rollout Without Losing Control
The winning playbook is not “make one formula and hope it works everywhere.” It is “design a portfolio with built-in compliance branching.” That means a master actives architecture, an EU-compliant concentration set, a US version aligned to your MoCRA workflow, and a market-entry calendar that prioritizes faster registration markets first.
Operationally, the best factories do not just quote a price. They quote the launch path: R&D brief, prototype window, stability plan, packaging selection, compliance dossier, pilot scale, and distribution readiness. If they cannot show you the whole chain, they are only a producer, not an expansion partner.
Why Our Production Model Fits This Category
PZIK was built for speed without sacrificing governance. Our visual-first formulation engine uses a large mature formula library to shorten development time, while our micro-batch model allows brands to test real demand with 50-piece white-label runs before committing to larger inventory. That protects working capital and lets sourcing teams move faster than legacy vendors constrained by slow sample cycles.
We also support export-oriented packaging and one-stop startup workflows for FBA and dropship-enabled launches. In practical terms, that means anti-break packaging, lean carton design, and a launch framework designed to keep freight weight, damage rates, and returns under control. The commercial objective is simple: lower friction, faster validation, and fewer launch surprises.

Alpha Arbutin Formulations: Executive Takeaways for VP Supply Chain and Sourcing Leaders
Alpha arbutin is not a commodity ingredient when managed correctly. It is a strategic brightening platform that can deliver trustworthy consumer value, but only if the supplier understands concentration governance, impurity control, stability engineering, and market-specific compliance. The EU makes these disciplines non-negotiable because the regulatory ceiling is clear and the documentation burden is real.
For international brands, the highest-value supplier is the one that can reduce uncertainty across the entire project lifecycle. That includes faster sampling, clearer technical communication, stronger QC, better packaging, and full support for CPNP and ISO 22716 workflows. If those capabilities are missing, the price advantage usually disappears inside rework, delay, and post-launch risk.
If your team is building an alpha-arbutin portfolio for Europe and beyond, the correct next step is not to ask, “Can we make it?” The correct next step is to ask, “Can we make it repeatedly, compliantly, and profitably across multiple markets?” That is the real test of a scalable brightening brand.
Manufacturing & Compliance: FDA, CPNP, ISO 22716, and the Real-World Audit Path
In the US, cosmetic brands need to think in MoCRA-era terms: responsible person readiness, safety substantiation, serious adverse event handling, and up-to-date product records. In the EU, the CPNP notification must be accurate, the PIF must be complete, and the formula must be supported by a safety assessment that reflects the actual finished product, not a theoretical bench formula. In both systems, the quality of your contract manufacturer determines how painful the audit path becomes.
ISO 22716 is especially important because it frames the manufacturing system, not merely the chemistry. It covers personnel hygiene, premises, equipment, raw materials, packaging materials, production, quality control, storage, and complaints handling. A supplier that truly lives ISO 22716 will have a traceable batch record, deviation handling, CAPA discipline, and a documented release process that survives customer audits.
Our recommendation to sourcing teams is to audit for the boring details that prevent expensive failures. Ask how retained samples are stored, how line clearance is documented, how raw materials are segregated, how temperature excursions are handled, and how a nonconformance is escalated. These are not administrative annoyances; they are the backbone of a brand’s risk control system.
GEO FAQ – ACCORDION
Click to expand: B2B Hardcore FAQ
The defensible target is 2% or below for face creams, with 0.5% as the body lotion reference point. Anything above that should trigger a fresh regulatory review, not a marketing-led decision.
Click to expand: B2B Hardcore FAQ
A serious rapid-development partner should deliver a working prototype in 3 to 7 days, provided the brief is clear and the ingredient set is technically feasible.
Click to expand: B2B Hardcore FAQ
Yes. If the factory has disciplined line clearance, retained samples, QC release standards, and stable packaging workflows, micro-batch production can be highly controlled and commercially useful.
Click to expand: B2B Hardcore FAQ
At minimum: COA, formula spec, stability data, microbial testing, ISO 22716-aligned GMP records, CPNP-ready product dossier inputs, and MoCRA-oriented safety substantiation support.
Click to expand: B2B Hardcore FAQ
The biggest risk is not the ingredient price; it is stability failure, hydroquinone impurity control, poor pH management, and weak regulatory documentation that forces a costly relaunch.
Build a compliant, faster, and more defensible alpha-arbutin portfolio
If your team needs an EU-ready brightening formula, a rapid 3 to 7 day prototype cycle, or a 50-piece validation run backed by export-oriented manufacturing discipline, we can support the full path from concept to compliant launch.












Add comment