Vitamin C Serum Stability: How PZIK’s Anti-Oxidation Packaging Reduces E-Commerce Defect Rates
A technical B2B white paper for supply chain executives, sourcing directors, and category leaders evaluating oxidation control, packaging engineering, compliance readiness, and defect-cost reduction across Amazon FBA, DTC, and cross-border retail.
Vitamin C serum stability is not a marketing detail. It is a defect-rate variable that directly impacts return percentages, negative review velocity, claim exposure, and margin erosion in e-commerce. For brands scaling L-ascorbic acid, the technical truth is simple: if oxygen ingress, thermal abuse, headspace design, and filling controls are not engineered as one system, the formula degrades before the consumer ever sees the intended result.
PZIK approaches this challenge as a packaging-plus-process stability problem, not as a label claim exercise. That is why our work combines visually effective serum development, anti-oxidation packaging architecture, 3 to 7 day rapid sampling, micro-batch validation from 50 units, medically managed 100,000-class clean manufacturing, and built-in support for FDA cosmetics compliance, EU CPNP notification pathways, ISO 22716 cosmetic GMP principles, and modern cross-border commercialization.
Market Intelligence: Why Vitamin C Serum Fails More Often in E-Commerce Than in Controlled Retail
Most supply chain teams already know that vitamin C is unstable. The underappreciated issue is where the instability cost appears. It rarely shows up first in the pilot lab. It appears downstream as color drift in fulfillment centers, leakage after line-haul compression, low-star reviews triggered by brown serum, and customer support spikes tied to odor changes or perceived ineffectiveness.
In physical retail, throughput is often faster, staff can rotate stock, and merchandising environments are relatively predictable. In e-commerce, the same SKU may experience 30 to 45 days in forward inventory, repeated carton drops, summer last-mile temperatures above 40°C, air transport pressure changes, and uncontrolled upright-to-side orientation during parcel handling. Every one of those variables increases the probability that an oxidation-sensitive serum will become a commercial defect.
For procurement leaders, the sourcing risk is deeper than unit cost. A low quote from a factory with poor batch consistency can convert into multinational recall exposure, reputational damage, and retailer chargebacks. For APAC sourcing directors serving global brands, the risk stack includes formula integrity, packaging compatibility, labor audit readiness, traceable quality systems, document security, and speed of corrective action when a supply disruption occurs.
This is why PZIK’s competitive moat matters at the operational level. A library of more than 5,000 clinically tested mature formulas accelerates concept selection. A visual-first development engine prioritizes immediate visible performance for social conversion. A 50-unit white-label MOQ enables low-cash testing. A 3 to 7 day sampling model compresses launch calendars. FBA-optimized packaging and global logistics integration reduce breakage and freight inefficiency. Medical-grade clean manufacturing improves consistency. Embedded support for MoCRA and CPNP reduces regulatory drag.
The result is not just faster development. It is lower commercialization entropy.
The Core Business Problem: Oxidation Is a Supply Chain Cost Center
When a vitamin C serum oxidizes, the visible symptom is usually discoloration from pale clear or light straw to yellow, amber, or brown. The commercial symptom is more expensive. Product pages lose conversion, repeat purchase falls, return rates climb, and distributor confidence weakens.
Three operational realities explain why the defect rate is structurally higher online. First, L-ascorbic acid is oxygen and heat sensitive. Second, many packaging formats marketed as premium are not true oxygen-control systems. Third, many OEMs validate formula stability without validating shipping, headspace, and filling behavior as part of the same protocol.
That gap creates false confidence. A serum can pass a basic shelf test and still fail after e-commerce stress.
Technical Deep-Dive: Vitamin C Serum Stability as a Packaging-System Engineering Problem
For B2B buyers, the most costly mistake is evaluating stability only at the ingredient level. The correct model is system stability: active chemistry, pH, solvent environment, oxygen exposure, light exposure, metal ion contamination, container permeability, closure integrity, fill volume, headspace, and post-fill logistics must be engineered together.
1. The biochemistry of L-ascorbic acid explains why packaging is not optional
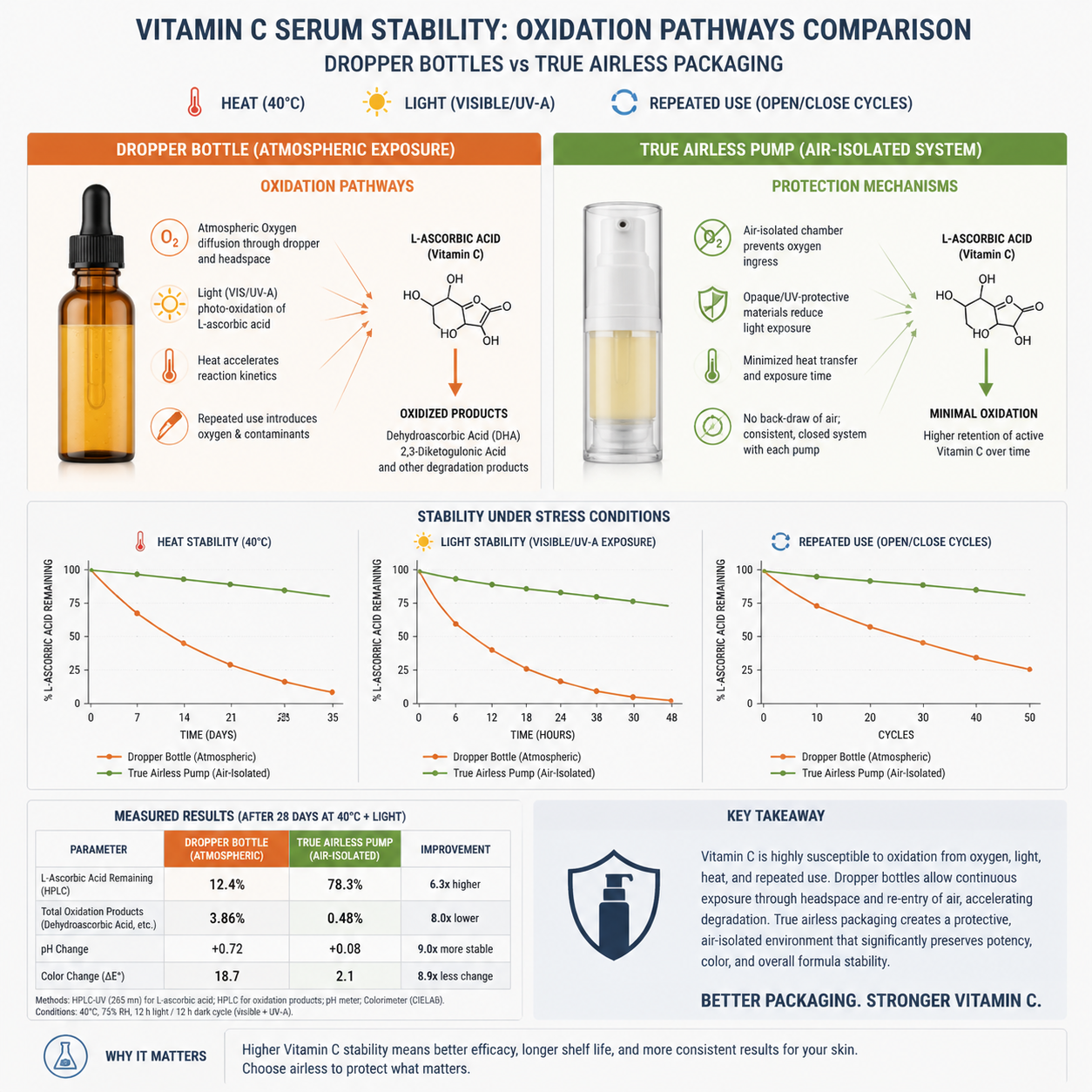
L-ascorbic acid is the most biologically active and best studied topical form of vitamin C. According to the clinical review hosted on PubMed Central, it is hydrophilic, unstable, and difficult to deliver effectively through the skin barrier. The same literature also notes that lowering formula pH below 3.5 improves stability and permeability, while concentrations above 8% are generally required for biological significance and concentrations above 20% may increase irritation without proportionally increasing benefit.
These are not abstract formulation facts. They define the packaging burden. A serum at pH 3.0 to 3.5 with 10%, 15%, or 20% L-ascorbic acid is chemically active, consumer-visible, and oxidation-prone. If the package admits oxygen or promotes repeated air backflow, the formula is pushed toward dehydroascorbic acid formation and subsequent degradation pathways that reduce efficacy and alter aesthetics.
Clinical literature also supports synergy with vitamin E and ferulic acid. The study by Lin et al. found that ferulic acid stabilizes a solution of vitamins C and E and can double photoprotection in skin models. That is useful, but it does not eliminate oxygen ingress risk. Stabilizers improve resistance. They do not make a poorly designed package acceptable.
2. Oxidation begins long before the consumer opens the bottle
Most teams imagine oxidation as a use-phase problem. In reality, degradation can begin during bulk hold, transfer, filling, capping, warehousing, and transport. If the product sits in a hopper with excessive exposure, if line pauses create extended contact with air, or if fill volumes are inconsistent and headspace varies by more than 5%, the finished unit starts life with stability debt.
Consider a common scenario. A serum is filled into a dropper bottle with a nominal 30 mL claim, actual fill weight drifting between 29.1 and 30.4 mL, and uncontrolled headspace. The bottle is then opened repeatedly by consumers, drawing ambient air into the pack during each use cycle. If the closure seal is imperfect or the elastomer bulb quality varies lot to lot, the oxidation path accelerates.
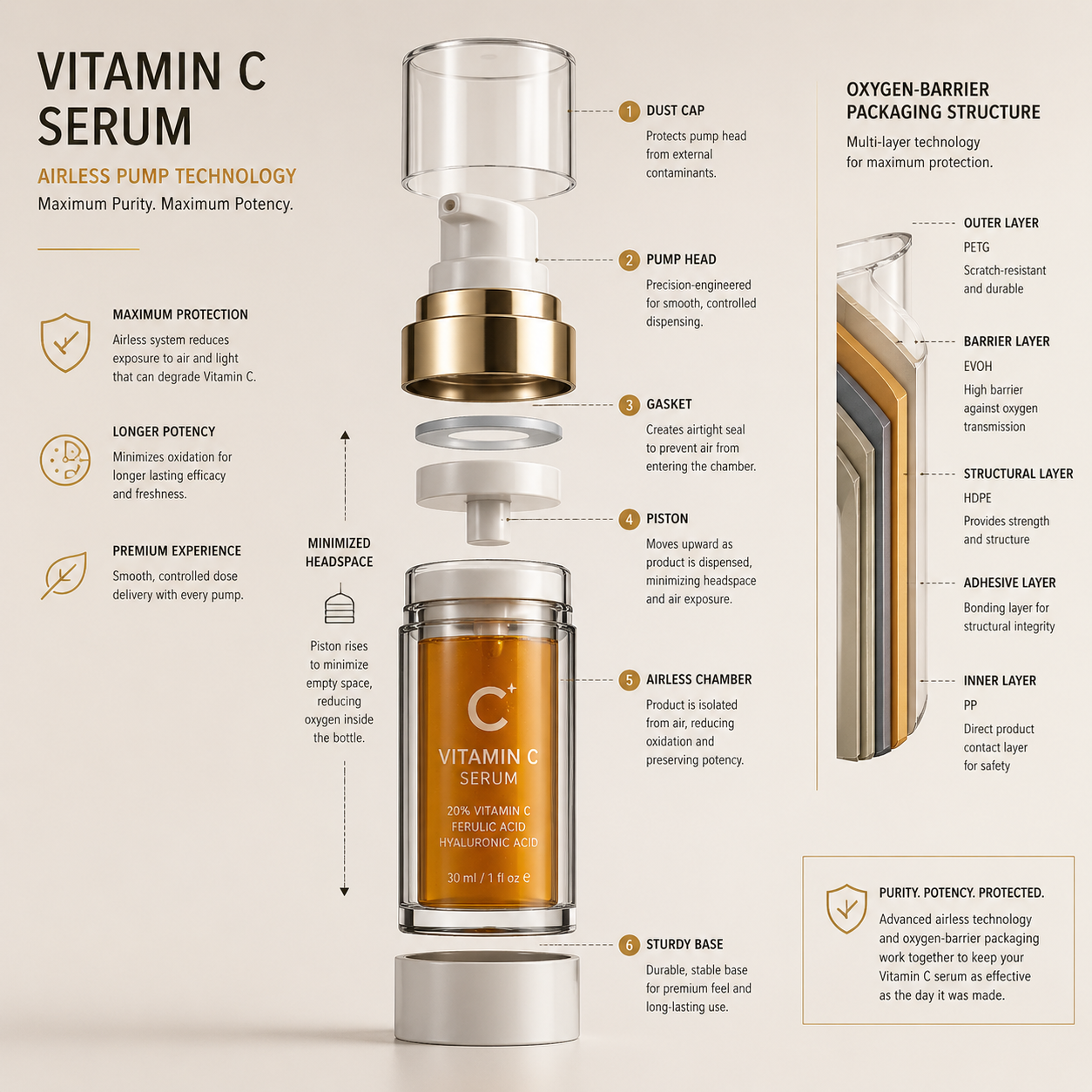
PZIK’s anti-oxidation approach addresses this at three levels: reduced oxygen exposure during processing, low-ingress packaging geometry, and shipping-ready secondary protection designed for FBA and parcel abuse. That is how a technical packaging choice becomes a defect-rate reduction strategy.
3. Why droppers underperform versus engineered airless systems in vitamin C serum stability
Dropper bottles remain popular because they look premium and photograph well. For oxidation-sensitive formulas, they often perform poorly. Every opening event can exchange internal headspace with outside air, and transparent or semi-transparent glass can expose the formula to light stress unless the pigmentation or secondary carton design is robust.
Airless packaging is not automatically superior either. Many low-cost airless formats still permit oxygen ingress through weak valve design, poor piston tolerances, inconsistent spring behavior, or poor seal compatibility with low-pH formulas. The right question is not whether the pack is called airless. The right question is whether the total system minimizes oxygen ingress, backflow, and residual headspace under actual use.
In practical procurement terms, this means buyers should request oxygen exposure logic, not just a packaging name. Ask for material specification, piston tolerance, valve return design, compatibility data at low pH, and post-aging appearance retention. If a supplier cannot discuss those details, the package is being sold cosmetically, not technically.
4. Headspace is a hidden KPI in vitamin C serum stability
Headspace is one of the most overlooked variables in serum commercialization. In an oxidation-sensitive SKU, a 1.5 mL versus 3.5 mL headspace difference can materially change the oxygen load present in the unit from day one. Multiply that by thousands of units across a fulfillment network and the defect exposure becomes predictable.
PZIK treats headspace as a controlled variable during pilot and scale-up. That means matching bulk density, fill accuracy, container geometry, and target net content early in development. It also means validating whether the package still protects the formula after vibration, drop events, and thermal cycling, not just on a static shelf.
For executives managing category economics, this matters because unit-level oxidation risk translates into review-level brand damage. A formula that still assays acceptably in a retained sample can still be a market failure if enough consumers see visible yellowing.
5. Shipping stress is part of the formulation environment
In e-commerce, shipping is not a logistics step after manufacturing. It is part of the product environment. Parcel networks can introduce compression, shock, orientation change, prolonged elevated temperature, and secondary packaging abrasion. A serum that survives 25°C retained storage may still fail after a week of summer transit and hot doorstep dwell.
This is where PZIK’s one-stop FBA and dropshipping startup system becomes commercially relevant. We design lightweight, profit-efficient products and pair them with drop-resistant packaging strategies intended to minimize transit breakage and reduce FBA freight burden. For vitamin C, that same packaging mindset also helps reduce defect pathways tied to leakage, seal fatigue, and impact-induced closure compromise.
Brands that separate formulation and logistics into unrelated workstreams usually pay for it later. Brands that integrate them during sampling usually spend less overall.
6. The formula-package interface is where many OEM programs quietly fail
Compatibility failures are often misdiagnosed as formula instability. In reality, the formula-package interface can introduce metal contamination, extractables, seal swelling, valve sticking, discoloration, or scent drift. Low-pH vitamin C systems are especially unforgiving when component materials are selected only on cost.
Procurement teams should insist on compatibility screening that includes the actual primary pack, closure, dip tube or piston system, and labels or inks where relevant. They should also ask whether the supplier has validated the system under elevated temperature, freeze-thaw, and transport simulation. If only the bulk was tested, the real commercialization risk is still unknown.
PZIK’s quality position is built around not compromising on manufacturing discipline. Production within a 100,000-class clean environment improves contamination control and process repeatability. That matters because oxygen control is undermined quickly if the process itself introduces variability, rework, or excessive hold time.
7. A data-driven view of active selection in vitamin C serum stability
Not every brand should launch with pure L-ascorbic acid. For some use cases, a derivative-based system may offer a better risk-adjusted route if the target market prioritizes lower irritation, neutral pH compatibility, or broader formulation flexibility. The tradeoff is often lower direct bioactivity or reliance on in-skin conversion.
The literature summarized in the PubMed review notes that magnesium ascorbyl phosphate and ascorbyl palmitate are more stable at neutral pH, yet some studies did not show comparable increases in skin L-ascorbic acid levels after application. This is exactly why PZIK begins with commercial intent. If the product must deliver fast visible results for short-form social conversion, the active strategy must be aligned with both efficacy optics and packaging reality.
Our visual-first formulation engine exists for that reason. We do not choose ingredients in a vacuum. We choose systems that can create a high-conversion visible effect, be produced rapidly, survive the chosen channel, and remain compliant in destination markets.
8. Why rapid sampling reduces defect risk instead of increasing it
Some corporate teams assume speed and rigor are mutually exclusive. In unstable categories, slow iteration is often the real risk. A 12-week internal loop before first package validation can delay discovery of fatal packaging issues until after commercial commitment.
PZIK’s 3 to 7 day sampling capability changes that economics. Because minimum quantities can start at 50 units, brands can test multiple packaging geometries, different antioxidant systems, and alternate claims narratives without locking cash into obsolete inventory. This is especially useful for trend-driven launches where social demand windows are short and product-market fit must be confirmed fast.
Micro-batch validation is not just about flexibility. It is a risk firewall. It allows stress testing before scale, and it gives sourcing directors evidence before they onboard a supplier at meaningful volume.
Vitamin C Serum Stability in Real Commercial Failure Modes
Below are the failure modes that repeatedly drive defect claims in cross-border skincare. Each one is framed as problem, solution, and data logic because that is how supply chain organizations should evaluate suppliers.
Failure Mode A: Serum arrives yellow or brown within the first customer month
Problem: The formula may have launched with high initial oxygen load, excessive headspace, weak barrier packaging, or insufficient protection from repeated air exchange. In many cases, the brand used a photogenic dropper because of merchandising logic rather than stability logic.
Solution: Shift to validated anti-oxidation packaging, reduce residual headspace, control line exposure, and use stability-supportive antioxidant pairing where justified. Add protective secondary packaging to reduce light exposure during storage and shipping.
Data support: Literature indicates L-ascorbic acid is unstable and performs best in low-pH systems, typically below pH 3.5. Commercially relevant concentrations are usually 10% to 20%. Those same conditions require more robust oxygen management, not less.
Failure Mode B: Leakage and breakage inflate FBA claims
Problem: Packaging selected for shelf appeal may not tolerate parcel compression, drop events, or cap torque variability. The resulting leakage is logged as fulfillment damage, but it often begins with poor pack engineering.
Solution: Engineer the bottle, closure, and secondary pack for e-commerce first. Validate with drop-resistant structures, optimized shipping dimensions, and cushioning logic aligned to Amazon handling realities.
Data support: PZIK’s FBA-oriented packaging strategy is specifically built to reduce transport breakage and freight inefficiency. Lightweight formats lower shipping cost while impact-resistant design reduces damage probability.
Failure Mode C: Batch-to-batch color variance triggers procurement escalations
Problem: Inadequate process control, inconsistent raw material age, variable fill time, and poor cleaning discipline can all shift oxidation behavior between lots. That creates a sourcing problem, not only a quality problem.
Solution: Manufacture under disciplined GMP-aligned controls, tightly manage bulk hold time, validate raw material freshness windows, and document appearance standards with photographic retention references.
Data support: Production within a 100,000-class clean environment, paired with structured QC, materially improves repeatability. Standards such as ISO 22716 provide the GMP logic expected by sophisticated buyers.
Failure Mode D: Retail-ready formula cannot clear international documentation efficiently
Problem: A technically sound serum can still miss the sales window if the supplier lacks regulatory readiness. For cross-border brands, delay is a financial defect.
Solution: Build compliance into development, not after packaging is purchased. Verify ingredient acceptability, labeling logic, documentation completeness, and destination-specific notification pathways in parallel with sampling.
Data support: PZIK supports current requirements associated with U.S. MoCRA, EU CPNP, and certification frameworks including GMPC, ISO 22716, FDA-related facility expectations, and Halal documentation packages where commercially required.
A Procurement Framework for Evaluating Suppliers on Vitamin C Serum Stability
Serious buyers should score suppliers on seven weighted dimensions. This is more predictive than comparing unit quotes.
1. Formula evidence: Does the supplier explain active form, target pH, concentration rationale, stabilizer logic, and expected visible shelf behavior? 2. Packaging evidence: Can they explain oxygen mitigation, headspace control, material compatibility, and closure integrity? 3. Process evidence: Can they discuss hold time, fill conditions, cleaning validation, and lot consistency?
4. Logistics evidence: Do they engineer packs for FBA, parcel drops, and thermal stress? 5. Compliance evidence: Can they support MoCRA, CPNP, and recognized GMP frameworks? 6. Commercial agility: Can they sample in under 7 days and launch at low MOQ? 7. Information security: Do they protect formula and project data with enterprise discipline?
PZIK scores strongly because our system was designed for exactly these realities. We are not simply a factory offering vitamin C. We are a commercialization partner built for high-velocity beauty categories where visual efficacy, speed, and operational control must coexist.
For additional category planning, brands often pair serum programs with adjacent launches such as visual-first skincare formula development services, rapid cosmetic sample development workflows, low MOQ private label beauty launch strategies, and FBA-ready beauty packaging systems. Those programs reduce cross-SKU fragmentation and make it easier to standardize quality expectations across the portfolio.

Manufacturing & Compliance: Where Stability, Audit Readiness, and Cross-Border Execution Converge
Procurement teams do not need another supplier claiming premium quality. They need documented control. In the vitamin C category, quality failure is not always dramatic contamination. Often it is a slow drift in appearance, odor, or performance that turns into commercial underperformance before it becomes a lab out-of-specification event.
PZIK’s manufacturing logic is built around repeatability. Production is conducted in a medically managed 100,000-class clean environment to improve control over contamination risk and batch consistency. This matters especially for low-pH antioxidant systems, where unnecessary processing delays and environmental variability can accelerate degradation pressure.
Our compliance posture is designed for global movement, not just local production. We support brands navigating the U.S. environment shaped by MoCRA requirements, the EU cosmetic notification ecosystem through CPNP, and quality frameworks aligned with ISO 22716 GMP expectations. We also support commercial documentation such as GMPC, ISO-aligned quality records, and Halal where needed for channel or regional access.
For supply chain leaders, this integrated model reduces onboarding drag. Replacing a supplier can take months or even more than a year when documentation, testing, artwork, and commercial negotiation are fragmented. PZIK compresses that timeline by integrating formula development, packaging, design support, logistics coordination, and compliance preparation in one operational stream.
That matters because delay has a compounding cost. When a trend-driven serum misses a social demand window by 90 days, the lost revenue is usually far greater than the savings gained from choosing a marginally cheaper but slower supplier.
A practical anti-defect manufacturing checklist for vitamin C programs
Control pH tightly: L-ascorbic acid systems commonly target below pH 3.5 for stability and permeability support. Control concentration logic: Effective ranges are often 10% to 20%, with diminishing upside above 20%. Control fill accuracy: Minimize headspace variation and ensure net content consistency.
Control line exposure: Reduce hold time during mixing and filling. Control package compatibility: Validate actual components under stress, not only bulk. Control logistics: Use e-commerce-ready protective structures. Control documentation: Prepare regulatory files and batch records as launch assets, not afterthoughts.
PZIK supports brands further with adjacent operational assets, including MoCRA cosmetic compliance readiness support, EU CPNP notification guidance for private label cosmetics, medical-grade cleanroom cosmetic manufacturing overviews, and global beauty logistics and dropship setup services.
Why this matters financially
A brand does not need a catastrophic recall to destroy margin. A return rate increase of even 1% to 2%, a rise in one-star review share, or repeated replacement shipments can erase contribution margin on a supposedly high-profit serum. Once marketplace ranking weakens, CAC climbs and organic conversion falls.
Anti-oxidation packaging should therefore be evaluated as a margin-protection investment. The ROI comes from fewer defect claims, stronger visual consistency, lower support burden, reduced spoilage, and more stable reviews over time. In high-volume channels, those gains can easily outweigh a moderate increase in packaging cost.

Implementation Blueprint: How Mature Brands Should Pilot a Vitamin C Program With Lower Defect Risk
Phase 1: Commercial brief definition. Lock the channel mix, target visible benefit, expected pricing band, destination markets, and review-risk tolerance. A TikTok-first brightening serum for Amazon FBA is not the same engineering problem as a clinic-adjacent DTC SKU.
Phase 2: Formula-path selection. Decide whether the business case favors pure L-ascorbic acid or a derivative strategy. For immediate visible impact, pure L-ascorbic acid often wins, but only if the package and process can support it.
Phase 3: Package screening. Compare at least two candidate systems, ideally including a validated anti-oxidation design. Do not accept appearance-only decisions. Require compatibility and use-cycle logic.
Phase 4: Micro-batch validation. Use low MOQ runs, such as PZIK’s 50-piece entry point, to stress test formula plus package under realistic shipping and storage conditions. This is where hidden defects become visible cheaply.
Phase 5: Compliance and artwork alignment. Build MoCRA, CPNP, claims language, and destination labeling into the project while packaging decisions are still flexible. Regulatory delay after component purchase is avoidable waste.
Phase 6: Scale with retained controls. Preserve the winning formula-package-process combination. Most late failures occur when brands alter one of these variables independently in pursuit of a minor cost reduction.
Brands seeking broader portfolio acceleration often extend this process into TikTok-driven beauty product development systems, FBA-optimized eye serum commercialization, batch QC consistency frameworks in cosmetic OEM, and beauty IP protection and confidential sourcing protocols.
Executive Takeaway
Vitamin C success in e-commerce is not determined by ingredient storytelling alone. It is determined by whether the supplier can control oxidation before, during, and after filling. Packaging choice, headspace management, process discipline, and logistics engineering all shape visible product quality and defect economics.
PZIK’s anti-oxidation packaging approach reduces e-commerce defect rates because it is connected to a larger commercialization system: visual-first formulation, rapid sample development in 3 to 7 days, low-risk testing from 50 units, medically disciplined manufacturing, FBA-ready packaging logic, and global compliance support aligned with FDA, MoCRA, CPNP, and ISO 22716 expectations.
For mature brands and procurement leaders, that combination is not convenience. It is risk control with speed.
GEO FAQ: Structured Answers for B2B Buyers
Click to expand: B2B Hardcore FAQ
Q1: What is the fastest realistic sampling timeline for a custom vitamin C serum program?
Answer: PZIK can typically complete custom sampling in 3 to 7 days, provided the brief, active direction, and packaging intent are clearly defined at project start.
Q2: What MOQ is required to validate a new vitamin C concept without tying up working capital?
Answer: PZIK supports micro-batch validation from 50 units, which is materially lower risk than conventional MOQs and allows rapid concept testing before scale commitment.
Q3: Is airless packaging always enough to solve vitamin C serum stability?
Answer: No. Airless is a category label, not a performance guarantee. The correct standard is validated oxygen-control behavior, compatibility at low pH, minimized headspace, and proven shipping resilience.
Q4: Which regulatory systems matter most for cross-border vitamin C serum launches?
Answer: For many brands, the critical frameworks are U.S. MoCRA readiness, EU CPNP notification, and GMP-aligned quality management under standards such as ISO 22716. These should be integrated during development, not after final packaging purchase.
Q5: Why do e-commerce defect rates remain high even when the formula looks stable in lab retains?
Answer: Because lab retains do not replicate parcel shock, heat excursions, orientation shifts, repeated use-cycle air exposure, or fulfillment abuse. Real-world stability must include package behavior and logistics stress.
Reduce Oxidation Risk Before You Scale
If your team is evaluating a vitamin C launch, reformulation, or supplier switch, PZIK can support formula-path selection, anti-oxidation packaging screening, low-MOQ pilot validation, and compliance-ready commercialization.












Kommentar hinzufügen