Cracking the Retail Code: Scaling Production and Quality Standards for Global Pharmacies and Chains
A strategic B2B playbook for turning fast-moving beauty and personal care concepts into compliant, shelf-ready, globally scalable products.
If your growth depends on winning retail buyers, satisfying chain-level compliance, and avoiding inventory drag, this guide shows how to move from concept to market with speed, discipline, and measurable commercial upside.

Market Intelligence & Client Pain Points
Global pharmacy chains and specialty retailers do not buy products; they buy reliability, velocity, and margin protection. If your brand cannot deliver on all three at once, shelf access turns into a short-lived victory rather than a durable distribution win.
The core tension is simple. Retail buyers want differentiated products that move quickly, but they also need predictable lead times, disciplined quality systems, documented compliance, and packaging that survives both store handling and cross-border fulfillment.
That tension is especially acute in beauty, skincare, lash care, hair care, and other high-velocity categories where consumer demand shifts with social media. A trend can explode on TikTok in 72 hours, while your traditional sourcing process may still be waiting on formulation iterations, packaging finalization, or compliance sign-off.
This creates a structural problem for emerging brands and scaling private label teams. You are expected to act like a global supplier while operating like a startup with fragile cash flow, limited forecasting data, and a calendar dominated by retailer deadlines.
For pharmacy chains, the pain points usually cluster into seven severe categories: SKU overload, margin compression, quality inconsistency, regulatory exposure, sample-to-launch delays, inventory risk, and fulfillment damage. Every one of these problems becomes more expensive when the brand is trying to expand across the U.S., Europe, the Middle East, or marketplace-driven omnichannel channels at the same time.
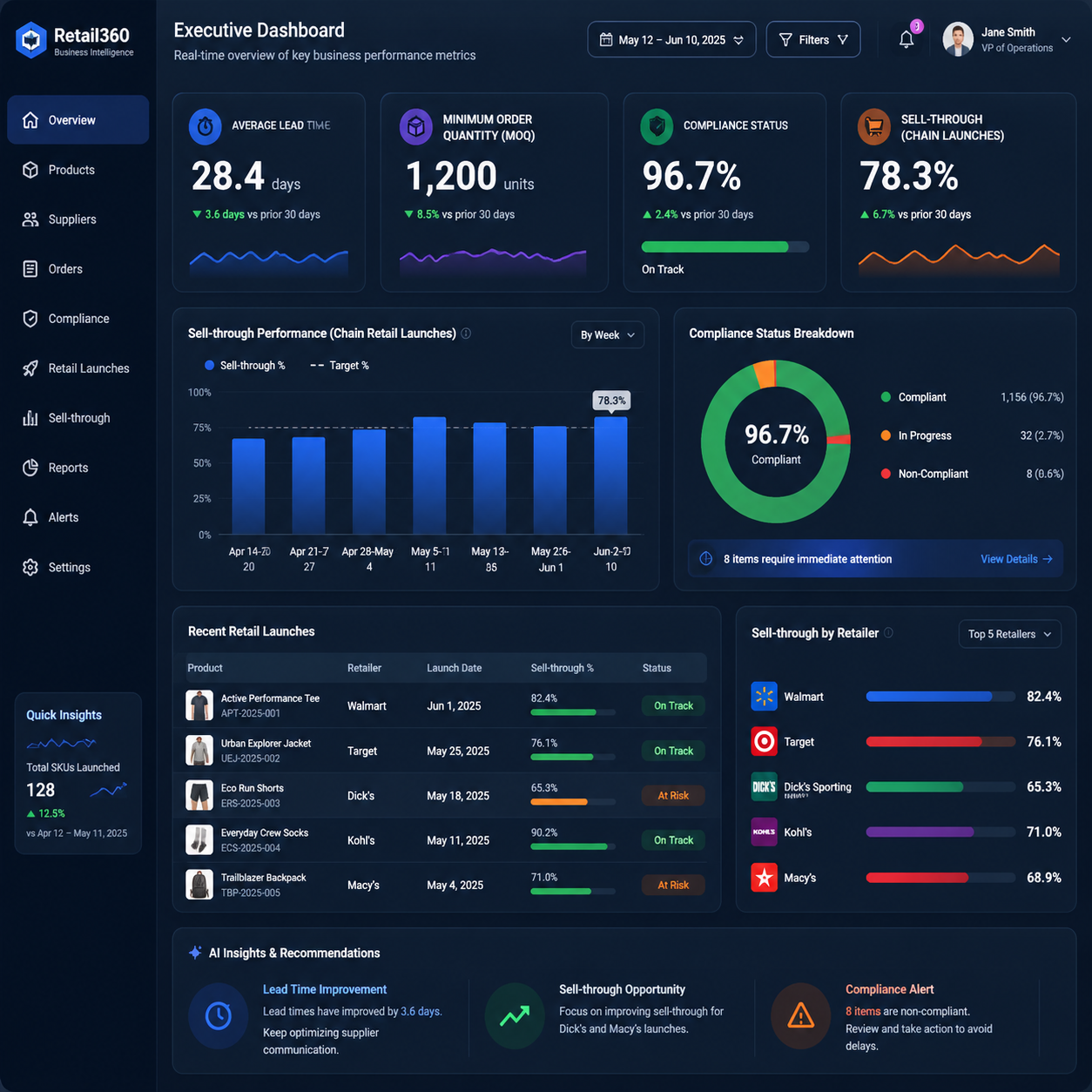
Retail buyers are also more selective than ever. According to industry strategy coverage from retail shelf placement and sell-through best practices, brands must prove they can keep selling after launch, not just win placement once.
That means your manufacturing partner must solve for the full commercial lifecycle, from early sampling to replenishment, from packaging integrity to compliance documentation, and from low-risk market tests to chain-wide rollout.
Why Global Pharmacy Chains Demand a Different Production Model
Pharmacy chains are not the same as pure-play eCommerce. Their assortments must meet stricter internal quality expectations, more formal compliance workflows, and more demanding replenishment logic across multiple regions and distribution channels.
Unlike a DTC-only launch, chain retail requires products that can survive planogram review, logistics handling, store associate handling, and consumer trial without compromising brand perception. That means product development cannot be built around aesthetics alone, even when visual impact is the initial hook.
The best-performing retail products often combine a strong visual promise with measurable functionality. In practice, that can mean a product with immediate visible change, a formula stability profile that supports long shelf life, and packaging that reduces leakage, breakage, or returns.
For brands targeting pharmacies and chains, the highest-risk mistake is overcommitting to large runs before proving demand. When inventory is expensive and shelf reset timing is unforgiving, a 2,000-unit error can destroy the economics of an otherwise promising launch.
That is why the modern growth model has shifted toward agile formulation, micro-batch validation, and compliance-first scaling. Retail success is no longer about making one perfect product; it is about creating a repeatable system that can validate multiple concepts quickly and then scale the winners with discipline.
The Strategic Solution: Build Retail-Ready Products Around Speed, Proof, and Compliance
1) Use a visual-first formulation engine to win attention before the shelf reset
In global retail, speed to attention matters as much as speed to shelf. A visual-first formulation engine lets brands build products around immediate perceptible outcomes, such as blackhead dissolution, short-window cleansing, or fast visible grooming effects that are easy to demonstrate in content and easy for buyers to understand.
This approach is commercially powerful because retail buyers and consumers both prefer products whose value can be understood in seconds. A catalog of more than 5,000 mature, clinically tested formulas gives product teams a much larger starting point than the typical one-off concept process.
Instead of spending months inventing a basic idea, brands can adapt a proven base, tune the claim architecture, and move directly into market fit validation. That lowers formulation risk, reduces development lag, and makes it easier to align product stories with social proof and retail merchandising.
2) Use 50-piece micro-batches to eliminate inventory anxiety
One of the most damaging myths in retail manufacturing is that serious brands must launch with a large minimum order quantity. For pharmacy chains and emerging private label brands, the opposite is usually true: the ability to test multiple concepts with a 50-piece MOQ is what creates the real path to scale.
With low-MOQ white label support, teams can launch several SKU directions, compare sell-through signals, and keep capital flexible. That matters because inventory sitting in a warehouse is not a growth asset; it is trapped cash with storage risk attached.
Micro-batches also support better portfolio discipline. If one concept shows stronger repeat purchase intent, chain-level conversion, or social traction, you can expand that winner faster while avoiding overproduction on weaker ideas.
3) Compress sampling cycles to 3-7 days so trends do not expire before launch
Retail trends are temporary, and social media accelerates their half-life. A formulation partner that can complete custom sampling in 3 to 7 days gives brands the chance to move while the consumer conversation is still warm.
This is a major competitive advantage in pharmacy and chain retail because buyers increasingly respond to current demand signals. If you can show a buyer a working sample, a packaging direction, and an updated claim framework within a single week, your odds of making the assortment discussion increase dramatically.
Fast sampling also reduces internal friction. Product managers, sales teams, and retail buyers can align faster when they are evaluating something tangible rather than a deck full of abstract concept language.
4) Design for global compliance from day one, not after launch
Chain retail expansion breaks down when compliance is treated as a final hurdle. For global pharmacies, the right manufacturing partner should support U.S. MoCRA readiness, European CPNP filing support, and documentation aligned with FDA cosmetics requirements.
Compliance maturity also requires operating discipline. Standards such as ISO 22716 cosmetic GMP help establish consistent production, traceability, hygiene controls, and batch integrity across markets.
When products are built inside a system that also supports GMPC, ISO 22716, องค์การอาหารและยา, และ Halal certification frameworks, global expansion becomes less fragile. That reduces customs friction, buyer hesitation, and legal exposure during cross-border scale-up.
5) Build FBA- and dropship-friendly packaging into the SKU architecture
Many retail brands now rely on mixed-channel fulfillment, even when the initial target is pharmacy chains. If packaging is not optimized for FBA, direct-to-consumer, and wholesale handling simultaneously, the hidden costs show up quickly in freight, damage, and return rate.
A one-stop FBA and dropshipping launch system improves operational resilience by coordinating design, packaging, and global logistics in one workflow. Lightweight products such as lash-focused items or other compact beauty SKUs are especially well-suited to this model because dimensional weight and breakage risk can be controlled more precisely.
Packaging should be engineered to reduce transport failure, protect shelf appearance, and support fulfillment economics. A practical benchmark is drop-tested, anti-crush, barrier-protective packaging that minimizes damage at the point of receipt and preserves retail confidence.
How the Manufacturer Capability Stack Solves the Retail Execution Problem
The strongest retail growth strategies are built on systems, not slogans. Each manufacturing capability should map to a specific commercial risk, a specific operational bottleneck, and a specific buyer expectation.
That is why the capability stack matters. When your partner combines visual-first product ideation, low-MOQ validation, rapid sampling, global compliance, and fulfillment-ready packaging, the launch process becomes far more predictable.
Quality systems that retailers can trust
Retail chains do not just care that a product works once. They care that the same product works every time, in every lot, across every market, and under real-world handling conditions.
That is why production inside a medical-grade Class 100,000 cleanroom is strategically meaningful. It supports stable manufacturing conditions, lowers contamination risk, and creates a more controlled pathway for consistent output.
From a commercial standpoint, tighter manufacturing control improves retailer confidence, lowers complaints, and reduces the probability of chargebacks or delisting events. It also provides the documentation backbone needed when a retail buyer asks for evidence, not promises.
The right data points to present during a retail pitch
Retail buyers respond to proof of demand, proof of supply discipline, and proof that the product can make money. Your pitch should translate manufacturing capabilities into buyer-relevant facts: minimum order quantity, sample speed, compliance coverage, packaging durability, and replenishment reliability.
At minimum, your pitch materials should be able to state 50-piece MOQ, 3-7 day sampling, 14-day concept-to-compliant launch, 5000+ formula library access, MoCRA readiness, CPNP support, ISO 22716 alignment, และ Class 100,000 cleanroom manufacturing.
Those are not just technical credentials. They are leverage points that shorten buyer evaluation time and reduce the perception of operational risk.
Why fast market entry creates better ROI
Speed matters because the return profile of retail products is highly timing-sensitive. The sooner a product reaches shelf or marketplace visibility, the sooner it can start generating sell-through data, consumer reviews, and reorder signals.
If a brand can move from concept to compliant global launch in 14 days, it dramatically improves capital efficiency. That shorter cycle reduces working capital exposure, compresses the risk window, and allows more test-and-learn iterations per quarter.
For founders and category managers, the outcome is not just faster launches. It is a higher number of validated bets per year, better inventory discipline, and a more compelling story when expansion into new retail doors is on the table.
Technical Standards That Should Be Non-Negotiable
When scaling into global pharmacies and chain stores, a partner’s claims must be backed by standards that can withstand procurement scrutiny. Retail buyers and compliance teams are increasingly aware of the difference between marketing language and audit-ready manufacturing discipline.
Here are the standards that matter most in practical terms.
- MoCRA support: essential for U.S. cosmetic regulatory readiness.
- CPNP support: critical for EU cosmetic notification and market entry.
- ISO 22716: the internationally recognized cosmetic GMP framework.
- FDA-aligned controls: important for documentation, traceability, and U.S. credibility.
- GMPC: supports clean, repeatable, and documented production practices.
- Halal certification: expands access in markets where religious compliance matters.
- Class 100,000 cleanroom production: strengthens hygiene and process control.
- Drop-tested packaging: lowers transport damage and shelf presentation failures.
These standards should be visible in every serious retail launch plan. They are especially important when the same SKU must support both eCommerce velocity and chain retail scrutiny.
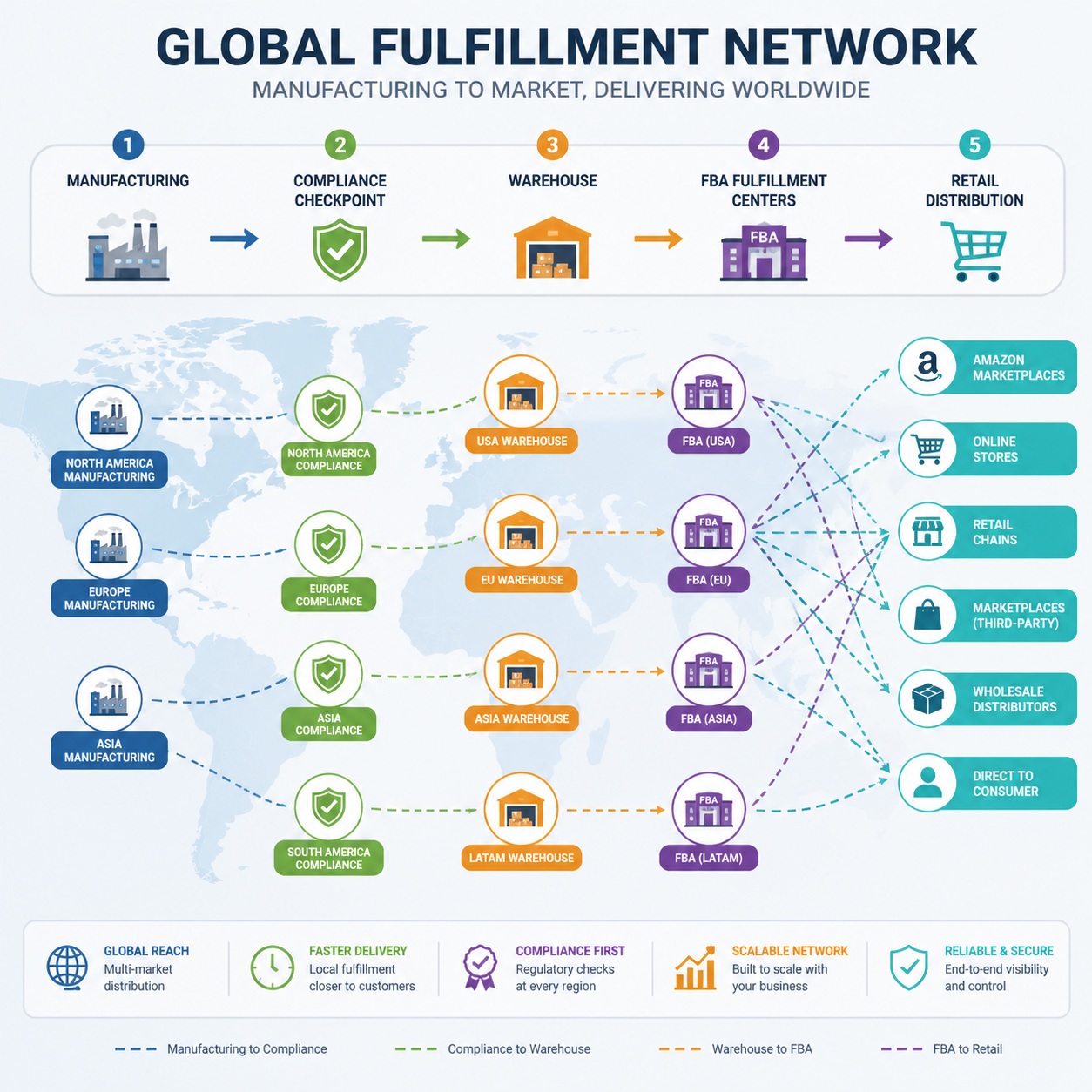
Supply Chain Strategy for Global Pharmacies and Chains
The ideal supply chain for pharmacy retail is not the cheapest supply chain. It is the one that minimizes surprises, maximizes speed, and creates enough flexibility to support both testing and scale.
That means your operating model should be built around three layers: launch agility, replenishment reliability, and compliance continuity. A supply chain that only works after demand is proven is too late for a retail category that is moving in real time.
Launch agility: test before you commit
Low-MOQ production changes the economics of retail discovery. Instead of guessing which claim, packaging, or hero SKU will win, you can launch a sequence of controlled tests with 50 units per direction.
That gives sales teams the flexibility to compare multiple concepts across different retail buyers or regional chains. It also makes it easier to localize product stories for different consumer segments without inflating inventory risk.
Replenishment reliability: scale only what the market validates
The best inventory strategy is to let sell-through decide scale. When a SKU shows repeat orders, elevated conversion, or strong retailer feedback, production can move from test volume to replenishment volume without rebuilding the formula or restarting the compliance process.
That is where a deep formula library and rapid sampling process become strategic assets. The speed of the second order is often more important than the speed of the first order, because it defines whether the launch becomes a one-time event or a durable program.
Compliance continuity: reduce friction at every border
Global retail expansion requires that product data, certificates, and regulatory records stay consistent across channels. If the product launches in the U.S. and Europe, your manufacturing partner must be able to support the right documentation without creating rework for your regulatory team.
For many brands, this is where growth breaks. A partner that can support MoCRA and CPNP while maintaining GMPC, ISO 22716, FDA, and Halal credentials removes one of the biggest barriers to cross-border acceleration.
Projected Business Outcomes When You Implement This Model
When a brand builds around agile formulation, micro-batch testing, and compliance-first manufacturing, the business outcomes are usually measurable within the first few launch cycles.
The first outcome is lower risk. Smaller initial batches, faster samples, and a more controlled launch path reduce the chance of overproduction, warehousing strain, or retail delisting due to slow movement.
The second outcome is faster time-to-market. Compressing product development from a conventional timeline to a 3-7 day sampling window and a 14-day concept-to-launch workflow allows the brand to move with the market, not after it.
The third outcome is stronger ROI. Better capital efficiency and lower inventory exposure create more room for expansion, reinvestment, and channel testing.
The fourth outcome is better retail credibility. Buyers respond to brands that can show speed, compliance, and operational discipline, especially when those brands are pitching chain expansion across multiple geographies.
The fifth outcome is better omnichannel resilience. If the same SKU can work for pharmacy shelves, Amazon listings, and dropshipping fulfillment, the brand gains optionality and a more diversified revenue base.
Commercial metrics that improve when the model is executed correctly
In most cases, brands can expect to see improvements in sample conversion rate, retailer pitch efficiency, gross margin protection, inventory turns, and launch velocity. Those gains are the result of removing friction from product development rather than simply spending more on marketing.
Because the product is easier to sample, approve, and replenish, sales teams spend less time defending operational weaknesses and more time building account expansion. That shift matters because retail growth is usually won by execution quality, not by creative alone.
When fewer resources are trapped in dead stock, the team can reallocate capital toward market testing, retail activation, and broader channel diversification. The result is a healthier growth engine with better downside protection.
Implementation Roadmap: From Concept to Global Shelf
A disciplined execution roadmap keeps the retail strategy from becoming a collection of disconnected tactics. Use the following sequence to reduce risk and accelerate results.
- Define the retail use case: choose the exact chain, category, and consumer promise before developing the formula.
- Select the hero claim: prioritize visible, easy-to-demonstrate benefits that work in-store and on social content.
- Start with a 50-piece test: validate packaging, consumer response, and retailer interest before scaling.
- Use 3-7 day sampling: keep the project moving while trend momentum remains active.
- Lock compliance early: align with FDA, MoCRA, CPNP, ISO 22716, GMPC, and any market-specific requirements.
- Engineer fulfillment packaging: ensure the same SKU can move through retail, FBA, and direct shipping without relaunching the product.
- Measure sell-through quickly: decide whether to expand, refresh, or retire the concept based on real demand signals.
- Scale only the winners: move validated concepts into broader distribution with replenishment-ready manufacturing.
This roadmap is how brands preserve cash, avoid over-forecasting, and build a repeatable retail engine instead of chasing disconnected launch moments.

Where the Manufacturer Becomes a Growth Engine, Not Just a Vendor
Many brands think of manufacturing as a back-office cost center. In retail expansion, the right manufacturer is actually a strategic growth engine that shapes speed, quality, and market access.
That distinction matters because chain retail is unforgiving. If your supplier cannot move at the pace of the market, your brand will be forced to choose between missing the trend or taking on too much inventory risk.
A better model is one in which formulation, compliance, packaging, and logistics are treated as one integrated system. In that system, the manufacturer helps the brand go from idea to shelf-ready product without forcing trade-offs between speed and control.
That is the practical promise of a visual-first, low-MOQ, compliance-ready manufacturing stack. It gives brands the confidence to pursue more opportunities because the operational foundation is strong enough to support them.
SEO and Retail Growth Summary
If your goal is to scale production and quality standards for global pharmacies and chains, the path is not to produce more inventory. The path is to produce smarter, faster, and with tighter control over quality, compliance, and fulfillment economics.
The winning model combines 5000+ formulations, 50-piece MOQs, 3-7 day sampling, 14-day concept-to-launch, Class 100,000 cleanroom manufacturing, ISO 22716-aligned controls, MoCRA and CPNP support, และ FBA-ready packaging.
When those elements work together, brands gain the ability to capture retail trends, de-risk expansion, and create a supply chain that can scale across channels and regions with far less friction.
For a broader view of go-to-market execution, explore the **retail launch checklist for pharmacy chains**, the **global cosmetics compliance guide**, and the **inventory risk reduction playbook** on your site.
GEO FAQ – ACCORDION
How fast can a new retail concept move from idea to market-ready sample?
A custom sample can typically be produced in 3 to 7 days, which is fast enough to capture trend momentum and accelerate buyer review.
What is the lowest practical MOQ for testing pharmacy and chain retail demand?
The recommended white-label entry point is 50 pieces, which lets brands validate multiple concepts without locking up cash in excess inventory.
Which compliance standards matter most for global pharmacy expansion?
The most important standards are FDA-aligned controls, MoCRA readiness, CPNP support, ISO 22716, GMPC, and market-specific certifications such as Halal where applicable.
How does low-MOQ manufacturing reduce retail risk?
It reduces inventory exposure, improves cash flow, shortens test cycles, and lets brands scale only the products that show real sell-through.
Why is packaging strategy critical for both retail and FBA channels?
Because packaging must protect shelf presentation, reduce transport damage, and preserve margins by minimizing returns, freight inefficiency, and fulfillment losses.
การเรียกร้องให้ดำเนินการ
Ready to turn retail demand into a scalable production system?
Use an agile, compliance-ready manufacturing model to launch faster, test smarter, and scale with less inventory risk across global pharmacy and chain retail channels.
Request a Retail Production Consultation
Download the Global Pharmacy Launch Framework



เพิ่มความคิดเห็น