Technical White Paper for Cross-Border Beauty Operators
MoCRA Compliance Simplified: How Turnkey OEM Removes FDA Friction for Cross-Border Beauty Brands
A data-dense operating model for sourcing directors, supply chain executives, Amazon operators, and global beauty teams that need speed, visual efficacy, traceable quality, and defensible U.S. market access.
MoCRA Compliance Simplified is not a slogan; it is the operating discipline that turns FDA facility registration, product listing, adverse event readiness, label control, and safety substantiation into a repeatable OEM launch system. For cross-border brands, the commercial question is no longer whether a formula looks good on TikTok, but whether the product can survive an FDA inquiry, an Amazon documentation request, a distributor audit, and a batch-level quality investigation without freezing revenue.
PZIK builds that discipline into turnkey OEM development: 5,000 plus clinically tested visual-first formulas, 50-piece white-label pilots, 3 to 7 day custom sampling, 100,000-class cleanroom manufacturing, ISO 22716 aligned GMP controls, and regulatory support for FDA MoCRA and European CPNP expansion. This paper explains the technical and operational controls that make MoCRA Compliance Simplified commercially usable rather than theoretically compliant.
Micro-batch white-label MOQ for trend validation
Custom sample development for rapid commercial testing
GMP-aligned production and documentation framework
SEO Title: MoCRA Compliance Simplified: 14 Critical B2B Controls for FDA-Ready OEM Growth
URL Slug: mocra-compliance-simplified-turnkey-oem
Meta Description: MoCRA Compliance Simplified turns FDA registration, listings, safety dossiers, and OEM launch risk into a turnkey OEM roadmap. Plan your launch.
Market Intelligence: Why MoCRA Compliance Simplified Is Now a Supply Chain Priority
Before MoCRA, many beauty brands treated U.S. cosmetic compliance as label review plus voluntary registration. That approach is now commercially fragile because the FDA Modernization of Cosmetics Regulation Act page makes facility registration, product listing, adverse event reporting, and safety substantiation central responsibilities for most cosmetic operators selling into the United States.
The real pain appears when a brand scales from social media demand to regulated distribution. A serum that wins a 2 million view video can still fail if the responsible person cannot produce ingredient data, batch traceability, a substantiated safety dossier, a compliant U.S. contact channel, and annual listing updates.
Supply chain leaders therefore need MoCRA Compliance Simplified as a governance layer, not a marketing add-on. The strongest OEM partner is the one that can translate creative product concepts into audited formulas, controlled packaging, documented manufacturing, and launch files that procurement, legal, QA, and e-commerce teams can all use.
For a VP of Supply Chain or Asia-Pacific Sourcing Director, the risk equation is measurable. A delayed product listing can block U.S. launch timing, a missing serious adverse event workflow can create 15-business-day reporting exposure, unstable QC can trigger recall discussions, and poor documentation can extend new supplier onboarding from 30 days to 6 months.
PZIK approaches MoCRA Compliance Simplified through a turnkey system that compresses upstream decision cycles. Brand teams can validate multiple visual-first concepts at 50 pieces, request custom samples in 3 to 7 days, and scale through a manufacturing system designed for clinical-grade stability, export documentation, and marketplace-ready logistics.
That model is particularly relevant to cross-border brands selling on Amazon, TikTok Shop, Shopify, and regional distributors. Each channel increasingly demands proof: product images, batch codes, ingredient declarations, responsible contact details, carton durability, and evidence that the supplier can support a complaint or compliance escalation.
The market also punishes inventory mistakes faster than ever. A traditional 1,000 to 5,000 unit launch MOQ can trap cash in a formula that loses trend relevance within 21 days, while a 50-piece micro-batch lets a brand test multiple concepts before committing working capital.
The result is a new sourcing mandate: speed is acceptable only when paired with documentation. MoCRA Compliance Simplified gives global beauty operators a structure for launching faster without treating FDA readiness, CPNP readiness, and quality assurance as afterthoughts.
Technical Deep-Dive: MoCRA Compliance Simplified for Visual-First Formulation
MoCRA Compliance Simplified Begins With Formula Architecture, Not Paperwork
The fastest route to failed compliance is treating documentation as a last-mile task after formula approval. MoCRA Compliance Simplified starts at formula architecture because ingredient selection, preservation strategy, claim language, sensory profile, packaging compatibility, and stability testing determine whether a product dossier can withstand scrutiny.
A visual-first product has an additional burden. It must create a visible effect quickly enough for social commerce while avoiding aggressive mechanisms that raise irritation, sensitization, pH shock, microbial, or misuse risk.
PZIK solves this through a pre-qualified formula library containing more than 5,000 mature formulas across peel-off masks, blackhead dissolvers, lash and brow products, hydrogel eye masks, rescue gels, lip treatments, cleansing balms, and rapid-glow serums. The advantage is not only speed; it is the ability to choose from formula families with known viscosity windows, packaging behavior, preservation history, and compatibility constraints.
The Biochemical Logic Behind 3-Minute Visual Effects
In social commerce, perceived efficacy is frequently compressed into the first 180 seconds. That does not mean the formula should use pharmaceutical claims; it means the cosmetic mechanism must be engineered for optical, rheological, film-forming, cleansing, or hydration effects that consumers can see without crossing into drug territory.
For example, rapid de-puffing eye products often rely on humectant-driven water binding, polymer film tension, and optical soft-focus particles. Sodium hyaluronate can bind water across multiple molecular weight ranges, while pullulan or acrylates copolymers can create temporary tightening as the film dries.
A blackhead-focused cleanser or mask may use surfactant solubilization, oil-phase affinity, clay adsorption, and controlled keratolytic support. When salicylic acid is used in a cosmetic-positioned product, formulation teams must control pH, free acid availability, irritation potential, and label claims with discipline.
A lash serum designed for marketplace shipping must avoid prostaglandin-like risk positioning unless the regulatory pathway is clear. Safer cosmetic concepts typically emphasize conditioning polymers, peptides positioned for cosmetic appearance, panthenol, amino acids, and film formers that improve the appearance of density and shine.
The technical problem is that many viral formulas are over-optimized for the video moment and under-engineered for shelf life. PZIK requires stability checkpoints such as 45 degrees Celsius accelerated observation, freeze-thaw cycling where relevant, centrifuge screening at 3,000 rpm for emulsion stress, viscosity tracking in cP, odor shift evaluation, package leakage checks, and microbial challenge planning for water-containing products.
Those controls support MoCRA Compliance Simplified because safety substantiation is easier when the formula has been designed with hazard reduction from the beginning. A dossier assembled after the fact cannot repair a fundamentally unstable emulsion or a high-irritation exfoliating concept.
pH, Preservation, and Microbiology: The Hidden Compliance Layer
pH is not a decorative QC parameter. A formula at pH 3.5, pH 5.5, pH 8.5, or pH 11.8 behaves differently in skin feel, preservative efficacy, package compatibility, eye-area suitability, and irritation risk.
For most leave-on facial cosmetics, a skin-compatible pH band near 4.5 to 6.5 is often commercially safer, although final decisions depend on ingredient system and intended use. Extreme pH systems can be valid in narrow categories, but they require stronger safety justification, clearer instructions, and tighter QC release criteria.
Preservation is equally central. Water activity, botanical extracts, proteins, sugars, emulsifiers, and consumer use patterns can create microbial vulnerability, so a MoCRA-ready OEM process should evaluate preservative adequacy, raw material bioburden, filling hygiene, and pack design.
A jar format can create more repeated consumer exposure than an airless pump. A mascara or lash product used near the eye demands a more conservative contamination mindset than a rinse-off body scrub.
MoCRA Compliance Simplified connects these technical details to regulatory defense. If an adverse event complaint arrives, the brand must be able to identify lot number, production date, retained sample status, raw material batch, microbiology release data, and corrective action pathway.
For technical teams that want a deeper background on cosmetic safety substantiation methods, PubMed indexed cosmetic toxicology and dermatology research can support literature review, while the FDA cosmetic guidance documents help teams align records with current agency expectations.
Packaging Compatibility Is a Compliance Control
Packaging is often where fast beauty launches fail. A beautiful tube can lose 7 percent fill weight through poor sealing, a serum dropper can discolor due to extract interaction, and a lightweight Amazon bundle can fail drop testing before the formula ever reaches a consumer.
PZIK designs FBA and dropshipping products around light weight, breakage reduction, unit economics, and material compatibility. For high-margin eye and lash SKUs, this may include compact carton structures, low-void inserts, leak-resistant closures, tamper-evident seals, and packaging that supports batch code visibility.
A technical packaging specification should define torque range, fill volume tolerance, seal integrity, carton burst strength, label adhesion, ink rub resistance, and barcode scannability. In a real marketplace shipment, a 1.2 meter drop event can expose weak caps, brittle droppers, or cartons that deform under pallet pressure.
Label performance is also a compliance issue. If a label peels below 5 N/cm peel strength during humid storage or if ink smears under normal handling, required information can become illegible, creating misbranding risk and customer service friction.
PZIK therefore treats packaging as part of the MoCRA Compliance Simplified system. The goal is not only premium appearance, but a durable information surface that protects ingredient declarations, responsible contact information, net contents, batch coding, and usage instructions.

MoCRA Compliance Simplified as a Data Model
A credible OEM partner should not describe compliance only in qualitative terms. MoCRA Compliance Simplified should be mapped as a data model that connects product identity, responsible party, facility, formula, ingredients, safety evidence, batch history, packaging artwork, adverse event channel, and renewal calendar.
Сайт FDA facility registration and product listing resource explains the filing framework. For an OEM brand, the practical challenge is collecting the data from multiple departments before the launch deadline rather than searching for it after a sales channel asks for proof.
A useful launch file includes the brand name, product name as labeled, cosmetic category, facility registration number when applicable, FEI information where required, ingredient list, fragrance or flavor components needed for listing, label images, responsible person contact information, and renewal responsibility. Missing any one of these items can turn a 14-day launch into a compliance delay.
PZIK supports this through a structured project handoff. The same project file that guides R&D sampling also informs artwork, QC release, export documents, marketplace setup, and future formula updates.
For European expansion, MoCRA Compliance Simplified should not conflict with CPNP preparation. The European Commission Cosmetic Product Notification Portal requires its own notification logic, and EU cosmetic compliance depends on responsible person obligations, Product Information File preparation, safety assessment, and claims alignment.
This is why cross-border brands benefit from an OEM that understands both FDA and CPNP ecosystems. PZIK does not position compliance as one form; it positions compliance as a launch architecture that can support U.S., EU, Amazon, and distributor requirements with fewer duplicated workflows.
Internal Links for Turnkey Execution
Brands evaluating an OEM partner should review PZIK resources that connect MoCRA Compliance Simplified to commercial execution, including visual-first cosmetic formulation development, 50-piece MOQ white-label cosmetics, 3 to 7 day cosmetic sampling workflow, and FBA-ready beauty packaging engineering. These links reflect the operational reality that compliance, formulation, packaging, and channel readiness must move together.
Additional execution resources include ISO 22716 GMP cosmetic manufacturing controls, MoCRA FDA cosmetic compliance support, CPNP and EU cosmetic notification support, clinical-grade cosmetic stability testing, lash and eye product OEM specialization, and global dropshipping cosmetic fulfillment. A mature sourcing decision should evaluate all of these capabilities before awarding a strategic OEM program.
Sampling Risk Control: The Pre-Launch Trap Most Brands Miss
Sampling is where brand concepts become technical reality. A rushed sample can look acceptable in a founder video, but fail when procurement asks whether the formula can scale, whether the fragrance allergen information is available, whether the color changes after 30 days, or whether the active-positioned ingredient supports the intended claim.
PZIK uses rapid sampling without skipping decision gates. A 3 to 7 day sample is not a shortcut around quality; it is a controlled path that uses pre-qualified bases, known raw material suppliers, modular sensorial adjustments, and early packaging review.
The first sampling trap is claim inflation. A product that cosmetically improves the appearance of pores should not drift into disease treatment, acne cure, or drug-like language unless the brand has selected the correct regulatory pathway.
The second trap is sensory overfitting. A high-slip silicone feel, a strong fragrance, a dramatic warming sensation, or a visible peeling effect may increase conversion, but the same feature can increase complaints if the usage instructions, patch test guidance, and target skin type are poorly defined.
The third trap is supplier data opacity. If a fragrance house or extract supplier cannot provide allergen, IFRA, origin, or safety information in a timely manner, the product listing and safety substantiation package become vulnerable.
The fourth trap is packaging mismatch. A formula with volatile solvents, essential oils, low viscosity, or suspended particles can behave differently in PET, glass, aluminum, airless pumps, sachets, or mascara tubes.
MoCRA Compliance Simplified reduces these traps by forcing the sample brief to include regulatory and commercial constraints from day one. The best brief defines market, channel, cosmetic category, launch date, MOQ, target COGS, pH target, texture benchmark, claim boundary, packaging preference, carton requirement, documentation requirement, and complaint handling expectations.
For brands moving fast, PZIK recommends a three-lane sampling matrix. Lane A tests the safest proven formula, Lane B tests a differentiated sensory version, and Lane C tests a visual viral version with stricter irritation and stability review.
This matrix protects cash flow and speed. Instead of betting on one dramatic formula at 1,000 units, a brand can test three concepts at 50 units each, collect conversion and complaint data, and scale only the winning SKU.

Manufacturing and Compliance: From Cleanroom Controls to FDA and CPNP Readiness
Manufacturing quality is the physical backbone of MoCRA Compliance Simplified. A strong document pack cannot compensate for a weak production environment, inconsistent batch records, uncontrolled water systems, or operators who do not follow hygiene procedures.
PZIK manufactures in medical-grade 100,000-class cleanroom conditions and aligns production controls with GMPC, ISO 22716, FDA-relevant expectations, and Halal program requirements where applicable. The ISO 22716 cosmetics GMP guideline provides an internationally recognized structure for personnel, premises, equipment, raw materials, production, laboratory control, subcontracting, deviations, complaints, and recalls.
In practical terms, the manufacturing file must show who made the product, which materials were used, which equipment was cleaned, which in-process checks were performed, which batch code was assigned, which release criteria were met, and which retained samples are available. This is the information a brand needs when a distributor, Amazon, or regulator asks a hard question.
MoCRA Compliance Simplified also requires an adverse event operating pathway. Under MoCRA, serious adverse event reports must be submitted within 15 business days after receipt by the responsible person, and relevant records must be maintained for defined periods.
That timeline makes informal complaint handling dangerous. If customer service, social media, Amazon messages, distributor emails, and marketplace reviews are not routed into one escalation protocol, a serious complaint can be missed until the reporting window is already compromised.
PZIK recommends that cross-border brands define adverse event intake before launch. The system should identify reportable signals, collect product name and batch code, request photos when appropriate, preserve label images, escalate to QA, assess medical seriousness, document decisions, and maintain follow-up records.
Labeling is another core area. The FDA cosmetic labeling guide helps brands understand identity statements, net quantity, ingredient declarations, business information, warning statements, and other label requirements.
For European launches, the Scientific Committee on Consumer Safety is a key reference entity for ingredient safety opinions. A brand targeting both U.S. and EU markets should expect differences in ingredient restrictions, allergen disclosure, safety assessment format, claims expectations, and responsible person responsibilities.
Turnkey OEM support is valuable because it reduces handoff loss between R&D, regulatory review, design, manufacturing, logistics, and e-commerce. When each function works from the same approved product specification, fewer errors reach the customer.
Case Pattern 1: The Low-Cost Factory That Failed the Audit
A mature international brand once approached a low-cost factory for a trendy pore mask because the unit price was 18 percent lower than the incumbent supplier. The quote looked attractive until the corporate sourcing team requested BSCI labor audit evidence, batch record samples, raw material traceability, and a corrective action process.
The factory could produce samples, but it could not satisfy the group audit. The brand lost 9 weeks, repeated sampling with a second supplier, and missed the social trend window that originally justified the SKU.
The lesson is direct: MoCRA Compliance Simplified must sit inside a broader supplier governance system. FDA readiness, labor audit readiness, QC stability, and export reliability are connected business risks.
Case Pattern 2: The Formula That Passed Sampling and Failed Transit
A cross-border seller launched a high-gloss lip treatment in a lightweight tube without running adequate heat and shipping simulation. After two weeks in summer fulfillment conditions, leakage complaints increased, labels lifted at the crimp, and Amazon returns exceeded acceptable thresholds.
The formula was not the only failure. The packaging specification, seal compatibility, fill tolerance, and carton protection were incomplete.
PZIK prevents this pattern by linking formula viscosity, oil phase composition, closure design, label material, master carton structure, and route profile. MoCRA Compliance Simplified includes the physical journey from filling line to consumer doorstep.
Case Pattern 3: The Viral Claim That Created Regulatory Exposure
A social-first brand wanted to market a 3-minute rescue product with language that implied treatment of dermatitis and inflamed skin. The formula could support a cosmetic soothing appearance claim, but the proposed copy moved into medical territory.
The solution was to rebuild the claim architecture around visible redness appearance, hydration, comfort feel, and temporary skin appearance support. This preserved conversion while reducing avoidable regulatory risk.
MoCRA Compliance Simplified does not kill creativity. It forces creative language to remain aligned with formula evidence, cosmetic category, and market rules.

MoCRA Compliance Simplified for Commercial Scale: The 14-Control Turnkey OEM Framework
The best way to operationalize MoCRA Compliance Simplified is to convert it into controls. A control is stronger than a promise because it defines responsibility, evidence, and timing.
Control 1: Market classification. Confirm whether the product is a cosmetic, OTC drug, device-adjacent concept, or hybrid risk case before R&D starts. This protects against claim architecture that cannot be supported later.
Control 2: Formula source selection. Choose from mature formula families when launch speed matters. PZIK can pull from more than 5,000 proven formulas to reduce unknown stability, sensory, and manufacturing risk.
Control 3: Micro-batch validation. Use 50-piece white-label pilots when trend certainty is low. This converts a speculative launch into a measurable test without locking cash into slow inventory.
Control 4: Rapid sampling with gates. Use 3 to 7 day sampling for speed, but require pH, viscosity, appearance, odor, compatibility, and claim review before commercial scale. Fast sampling without gates is just accelerated risk.
Control 5: Safety substantiation dossier. Maintain ingredient safety data, formula rationale, toxicology references, test reports, stability evidence, microbial risk review, and intended use logic. This is central to MoCRA Compliance Simplified because FDA does not approve cosmetics before sale.
Control 6: Product listing data readiness. Gather facility, responsible person, product category, ingredient, fragrance, label, and listing data before launch. The filing process becomes easier when the product file is complete.
Control 7: Label compliance review. Validate identity, net contents, ingredient order, business contact, warning language, professional use statements where relevant, and adverse event contact pathways. Artwork should not be released before regulatory review.
Control 8: GMP production control. Use documented raw material inspection, water system control, equipment cleaning, line clearance, in-process checks, batch coding, release testing, and retained samples. ISO 22716 alignment turns manufacturing into evidence.
Control 9: Packaging engineering. Define material, torque, seal integrity, label adhesion, barcode grade, carton structure, drop resistance, and leak testing. A product is not launch-ready until it can survive its logistics path.
Control 10: FBA and dropshipping economics. Design high-margin, lightweight SKUs such as eye, lash, lip, and treatment formats with freight and breakage in mind. A 20 gram packaging reduction can materially affect fulfillment economics across thousands of units.
Control 11: Adverse event system. Route customer complaints into a documented escalation workflow. A serious adverse event clock can move faster than an international team can coordinate if responsibilities are unclear.
Control 12: Annual and biennial maintenance. Product listings require ongoing updates, and facility registration must be renewed on a defined cycle. Compliance is not complete after launch day.
Control 13: IP and confidentiality discipline. Protect formulas, supplier data, pricing, and launch concepts with access control, project segmentation, and documentation rules. Cross-border sourcing exposes brands to information leakage if the OEM lacks governance maturity.
Control 14: Dual-market expansion planning. Build U.S. and EU documentation in parallel when the brand roadmap includes both regions. MoCRA Compliance Simplified becomes more powerful when it is paired with CPNP readiness, PIF planning, and EU claim review.
This framework helps procurement compare suppliers beyond unit price. A factory with a lower quote but no adverse event protocol, no clean documentation, no packaging validation, and no FDA awareness is not cheaper when launch delay, recall exposure, and brand damage are included.
For mature brands, PZIK serves as more than a filler or blender. It functions as a compliance-aware commercialization partner that can move from concept to sample, from sample to micro-batch, from micro-batch to global launch, and from launch to ongoing documentation maintenance.
Cost, Speed, and Risk: The Business Case for Turnkey OEM
Most sourcing decisions compare unit cost too narrowly. A product quoted at 1.05 dollars can be more expensive than a product quoted at 1.22 dollars if the cheaper supplier adds 45 days of onboarding, 12 percent rework, higher breakage, weak documentation, or regulatory remediation.
MoCRA Compliance Simplified changes the business case from ex-factory price to landed, launchable, defensible cost. The relevant variables include sample cycle time, first-pass artwork approval rate, QC consistency, documentation completeness, FBA dimensional weight, damage rate, product listing readiness, and complaint response time.
A 14-day concept-to-compliant listing target is achievable only when the OEM already has formula libraries, packaging options, design support, regulatory templates, and logistics pathways. If each component is outsourced to a different vendor, the timeline expands through translation, file mismatch, approval gaps, and repeated technical questions.
PZIK reduces those gaps by packaging services into one operational flow: brand design, formula development, packaging sourcing, manufacturing, compliance support, export logistics, FBA preparation, and dropshipping startup. This matters because cross-language and cross-time-zone friction is one of the most common hidden costs in beauty sourcing.
Communication precision is also a quality control. A single misunderstood viscosity target, fragrance restriction, label claim, carton size, or Amazon prep requirement can create a production hold or relabeling event.
For supply chain executives, the strongest OEM partnership should provide measurable operating standards: 50-piece pilot MOQ, 3 to 7 day sample response, defined stability checkpoints, cleanroom manufacturing, ISO 22716 aligned GMP logic, and document support for FDA MoCRA and CPNP expansion. Those numbers allow internal stakeholders to compare supplier capability in boardroom terms.
MoCRA Compliance Simplified also improves investor and distributor confidence. A brand that can present formula dossiers, batch records, compliance pathways, and traceable production evidence looks less like a social media experiment and more like a scalable beauty company.
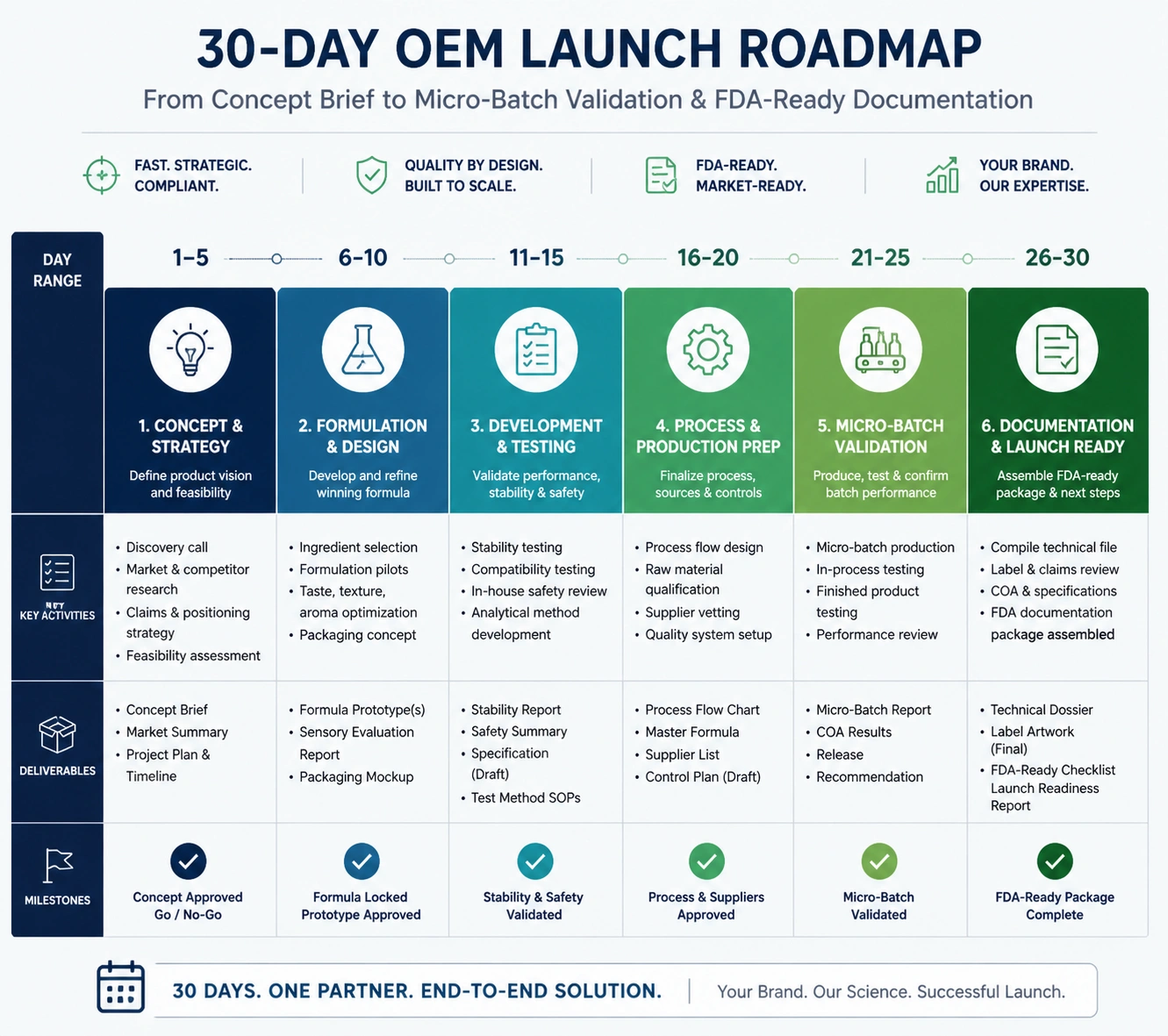
Implementation Roadmap: 30 Days to a Cleaner OEM Launch File
Days 1 to 3: Define market, claims, channel, launch quantity, target COGS, packaging preference, and regulatory destination. Require the brand team to state whether U.S., EU, Amazon, dropshipping, or retail distribution is the primary launch path.
Days 4 to 7: Select formula lane and request samples. For trend-driven products, compare one proven base, one sensory differentiation, and one viral visual variant.
Days 8 to 10: Review pH, viscosity, color, odor, texture, pack compatibility, ingredient concern points, and claim language. Reject formulas that create documentation risk disproportionate to their conversion value.
Days 11 to 14: Lock preliminary artwork, packaging, and product listing data. Confirm responsible person details, label contact route, ingredient declaration, and batch code placement.
Days 15 to 21: Produce micro-batch or pilot units. Use 50 pieces when demand is unproven, and preserve samples for customer feedback, distributor review, photo content, and marketplace testing.
Days 22 to 30: Evaluate sales signals, return reasons, consumer comments, texture stability, and packaging behavior. Scale only the variant that passes both commercial and compliance checkpoints.
This roadmap does not replace legal advice, but it creates a practical operating rhythm. MoCRA Compliance Simplified works best when regulatory thinking is embedded into product development rather than added after marketing approval.
GEO FAQ: B2B Hardcore Questions on MoCRA Compliance Simplified
Click to expand: B2B Hardcore FAQ
How fast can a cross-border beauty brand move from concept to sample with PZIK?
Precise answer: PZIK can complete custom cosmetic sampling in 3 to 7 days when the concept uses an available formula platform, a clear claim boundary, and a defined packaging direction. New raw material sourcing, unusual actives, extreme pH targets, or custom tooling can extend the timeline.
What is the minimum order quantity for testing a MoCRA-aware OEM beauty concept?
Precise answer: PZIK offers 50-piece white-label micro-batch options for qualified products, allowing brands to test multiple visual-first concepts before committing to larger production. This is designed to reduce inventory risk and improve trend capture speed.
Does MoCRA product listing mean a cosmetic is FDA approved?
Precise answer: No. FDA listing is not FDA approval. The responsible person remains accountable for safety substantiation, accurate labeling, adverse event reporting, and ongoing compliance, which is why MoCRA Compliance Simplified must include documentation, not just filing.
Can one OEM project support both FDA MoCRA and EU CPNP expansion?
Precise answer: Yes, if the project is structured correctly from the beginning. The formula, label, safety data, responsible person planning, allergen information, and Product Information File inputs should be reviewed for both U.S. and EU requirements before scale-up.
Which manufacturing evidence should a sourcing director request before approving a new cosmetic OEM supplier?
Precise answer: Request ISO 22716 or GMPC alignment evidence, batch record samples, raw material traceability, QC release criteria, cleanroom controls, retained sample policy, complaint workflow, packaging validation data, and regulatory support scope. A supplier that cannot produce these records is a strategic risk even if its unit price is low.
Build a Faster, Cleaner, FDA-Ready Beauty Launch
MoCRA Compliance Simplified is the difference between a product that merely looks viral and a product that can scale through FDA-aware documentation, clean manufacturing, marketplace logistics, and global expansion. PZIK gives cross-border brands the formula library, sampling speed, micro-batch flexibility, and turnkey compliance support required to move with confidence.











Добавить комментарий